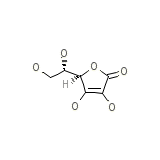
Natrascorb Injectable




Natrascorb Injectable Brand names, Natrascorb Injectable Analogs
- Vitamin C
- AA
- Adenex
- Allercorb
- Antiscorbic Vitamin
- Antiscorbutic Vitamin
- Arco-Cee
- Ascoltin
- Ascor-B.I.D.
- Ascorb
- Ascorbajen
- Ascorbate
- Ascorbic Acid
- Ascorbicab
- Ascorbicap
- Ascorbicin
- Ascorbin
- Ascorbutina
- Ascorin
- Ascorteal
- Ascorvit
- C-Level
- C-Long
- C-Quin
- C-Span
- C-Vimin
- Cantan
- Cantaxin
- Catavin C
- Ce Lent
- Cebicure
- Cebid
- Cebion
- Cebione
- Cecon
- Cee-Caps Td
- Cee-Vite
- Cegiolan
- Ceglion
- Celaskon
- Celin
- Cemagyl
- Cemill
- Cenetone
- Cenolate
- Cereon
- Cergona
- Cescorbat
- Cetamid
- Cetane
- Cetane-Caps Tc
- Cetane-Caps Td
- Cetebe
- Cetemican
- Cevalin
- Cevatine
- Cevex
- Cevi-Bid
- Cevimin
- Cevital
- Cevitamic Acid
- Cevitamin
- Cevitan
- Cevitex
- Cewin
- Ciamin
- Cipca
- Citriscorb
- Colascor
- Component of Chromagen
- Component of Cortalex
- Component of E and C-Level
- Component of Endoglobin Forte
- Component of Ferancee
- Component of Freancee
- Component of Stuartinic
- Component of Tolfrinic
- Component of Veliten
- Concemin
- Davitamon C
- Duoscorb
- Ferrous Ascorbate
- Hicee
- Hybrin
- IDO-C
- Iron-Ascorbic Acid Complexes
- Kyselina Askorbova
- L-Ascorbate
- L-Ascorbic Acid
- L-Ascorbic Acid Extra Pure
- L-Lyxoascorbic Acid
- L-Xyloascorbic Acid
- Laroscorbine
- Lemascorb
- Liqui-Cee
- Mediatric Component Of
- Meri-C
- Natrascorb
- Natrascorb Injectable
- Planavit C
- Proscorbin
- Redoxon
- Ribena
- Roscorbic
- Scorbacid
- Scorbu-C
- Secorbate
- Sodascorbate
- Testascorbic
- Vicelat
- Vicin
- Vicomin C
- Viforcit
- Viscorin
- Vitace
- Vitacee
- Vitacimin
- Vitacin
- Vitamisin
- Vitascorbol
- Xitix
Natrascorb Injectable Brand Names Mixture
- No information avaliable
Natrascorb Injectable Chemical_Formula
C6H8O6
Natrascorb Injectable RX_link
http://www.rxlist.com/cgi/generic3/vitamc.htm
Natrascorb Injectable fda sheet
Natrascorb Injectable msds (material safety sheet)
Natrascorb Injectable Synthesis Reference
No information avaliable
Natrascorb Injectable Molecular Weight
176.124 g/mol
Natrascorb Injectable Melting Point
191 oC
Natrascorb Injectable H2O Solubility
40 g/L
Natrascorb Injectable State
Solid
Natrascorb Injectable LogP
-1.305
Natrascorb Injectable Dosage Forms
Capsule; Tablet (chewable); Tablet (immediate release); Tablet (extended release); Lozenge; Solution; Parenteral injection
Natrascorb Injectable Indication
Used to treat vitamin C deficiency, scurvy, delayed wound and bone healing, urine acidification, and in general as an antioxidant. It has also been suggested to be an effective antiviral agent.
Natrascorb Injectable Pharmacology
Ascorbic Acid (vitamin C) is a water-soluble vitamin indicated for the prevention and treatment of scurvy, as ascorbic acid deficiency results in scurvy. Collagenous structures are primarily affected, and lesions develop in bones and blood vessels. Administration of ascorbic acid completely reverses the symptoms of ascorbic acid deficiency.
Natrascorb Injectable Absorption
70% to 90%
Natrascorb Injectable side effects and Toxicity
No information avaliable
Natrascorb Injectable Patient Information
No information avaliable
Natrascorb Injectable Organisms Affected
Humans and other mammals