Naptazane




Naptazane Brand names, Naptazane Analogs
Naptazane Brand Names Mixture
- No information avaliable
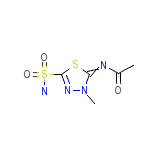
Naptazane Chemical_Formula
C5H8N4O3S2
Naptazane RX_link
http://www.rxlist.com/cgi/generic2/methaz.htm
Naptazane fda sheet
Naptazane msds (material safety sheet)
Naptazane Synthesis Reference
Young, et al., J. Am. Chem. Soc. 78, 4649 (1956)
Naptazane Molecular Weight
236.274 g/mol
Naptazane Melting Point
213.5 oC
Naptazane H2O Solubility
3500 mg/L
Naptazane State
Solid
Naptazane LogP
0.231
Naptazane Dosage Forms
Tablet
Naptazane Indication
For treatment of chronic open-angle glaucoma and acute angle-closure glaucoma
Naptazane Pharmacology
Methazolamide is topical carbonic anhydrase inhibitor. Methazolamide is indicated for the reduction of elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension who are insufficiently responsive to beta-blockers. Methazolamide is a sulfonamide derivative; however, it does not have any clinically significant antimicrobial properties. Although methazolamide achieves a high concentration in the cerebrospinal fluid, it is not-considered an effective anticonvulsant. Methazolamide has a weak and transient diuretic effect, therefore use results in an increase in urinary volume, with excretion of sodium, potassium and chloride.
Naptazane Absorption
Methazolamide is well absorbed from the gastrointestinal tract.
Naptazane side effects and Toxicity
Electrolyte imbalance, development of an acidotic state, and central nervous system effects might be expected to occur in the case of an overdose.
Naptazane Patient Information
Naptazane Organisms Affected
Humans and other mammals