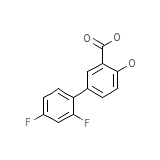
Dolobis




Dolobis Brand names, Dolobis Analogs
Dolobis Brand Names Mixture
- No information avaliable
Dolobis Chemical_Formula
C13H8F2O3
Dolobis RX_link
http://www.rxlist.com/cgi/generic3/diflunisal.htm
Dolobis fda sheet
Dolobis msds (material safety sheet)
Dolobis Synthesis Reference
No information avaliable
Dolobis Molecular Weight
250.198 g/mol
Dolobis Melting Point
210-221 oC
Dolobis H2O Solubility
Practically insoluble (14.5 mg/L) at neutral or acidic pH.
Dolobis State
Solid
Dolobis LogP
3.876
Dolobis Dosage Forms
Tablets for oral administration (250 mg or 500 mg)
Dolobis Indication
For acute or long-term use for symptomatic treatment of mild to moderate pain, osteoarthritis, and rheumatoid arthritis.
Dolobis Pharmacology
Diflunisal is a nonsteroidal drug with analgesic, anti-inflammatory and antipyretic properties. It is a peripherally-acting non-narcotic analgesic drug. Habituation, tolerance and addiction have not been reported. Diflunisal is a difluorophenyl derivative of salicylic acid. Chemically, diflunisal differs from aspirin (acetylsalicylic acid) in two respects. The first of these two is the presence of a difluorophenyl substituent at carbon 1. The second difference is the removal of the 0-acetyl group from the carbon 4 position. Diflunisal is not metabolized to salicylic acid, and the fluorine atoms are not displaced from the difluorophenyl ring structure.
Dolobis Absorption
Rapidly and completely absorbed following oral administration, with a bioavailability of 80-90%.
Dolobis side effects and Toxicity
Oral LD50 in rat, mouse, and rabbit is 392 mg/kg, 439 mg/kg, and 603 mg/kg, respectively. Symptoms of overdose include coma, tachycardia, stupor, and vomiting. The lowest dose without the presence of other medicines which caused death was 15 grams.
Dolobis Patient Information
Dolobis Organisms Affected
Humans and other mammals