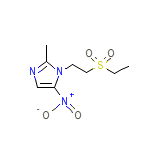
D,L-Tenofovir




D,L-Tenofovir Brand names, D,L-Tenofovir Analogs
D,L-Tenofovir Brand Names Mixture
- No information avaliable
D,L-Tenofovir Chemical_Formula
C8H13N3O4S
D,L-Tenofovir RX_link
No information avaliable
D,L-Tenofovir fda sheet
D,L-Tenofovir msds (material safety sheet)
D,L-Tenofovir Synthesis Reference
No information avaliable
D,L-Tenofovir Molecular Weight
247.273 g/mol
D,L-Tenofovir Melting Point
127-128 oC
D,L-Tenofovir H2O Solubility
1.99E+004 mg/L
D,L-Tenofovir State
Solid
D,L-Tenofovir LogP
0.119
D,L-Tenofovir Dosage Forms
Pink film-coated oral tablets contain 500 mg or 250 mg of tinidazole.
D,L-Tenofovir Indication
For the treatment of trichomoniasis caused by T. vaginalis in both female and male patients. Also for the treatment of giardiasis caused by G. duodenalis in both adults and pediatric patients older than three years of age and for the treatment of intestinal amebiasis and amebic liver abscess caused by E. histolytica in both adults and pediatric patients older than three years of age.
D,L-Tenofovir Pharmacology
Tinidazole is a synthetic antiprotozoal agent. Tinidazole demonstrates activity both in vitro and in clinical infections against the following protozoa: Trichomonas vaginalis, Giardia duodenalis (also termed G. lamblia), and Entamoeba histolytica. Tinidazole does not appear to have activity against most strains of vaginal lactobacilli.
D,L-Tenofovir Absorption
Rapidly and completely absorbed under fasting conditions. Administration with food results in a delay in Tmax of approximately 2 hours and a decline in Cmax of approximately 10% and an AUC of 901.6 ± 126.5 mcg hr/mL.
D,L-Tenofovir side effects and Toxicity
There are no reported overdoses with tinidazole in humans. In acute studies with mice and rats, the LD 50 for mice was generally > 3,600 mg/kg for oral administration and was > 2,300 mg/kg for intraperitoneal administration. In rats, the LD 50 was > 2,000 mg/kg for both oral and intraperitoneal administration.
D,L-Tenofovir Patient Information
D,L-Tenofovir Organisms Affected
Trichomonas vaginalis, Giardia duodenalis (also termed G. lamblia), and Entamoeba histolytica