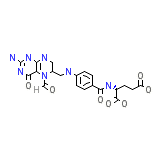
Citrovorum factor




Citrovorum factor Brand names, Citrovorum factor Analogs
Citrovorum factor Brand Names Mixture
- No information avaliable
Citrovorum factor Chemical_Formula
C20H23N7O7
Citrovorum factor RX_link
http://www.rxlist.com/cgi/generic2/leucovorin.htm
Citrovorum factor fda sheet
Citrovorum factor msds (material safety sheet)
Citrovorum factor Synthesis Reference
No information avaliable
Citrovorum factor Molecular Weight
473.44 g/mol
Citrovorum factor Melting Point
No information avaliable
Citrovorum factor H2O Solubility
Complete
Citrovorum factor State
Solid
Citrovorum factor LogP
-1.744
Citrovorum factor Dosage Forms
Liquid; Powder for solution; Solution; Tablet
Citrovorum factor Indication
For the treatment of osteosarcoma (after high dose methotrexate therapy). Used to diminish the toxicity and counteract the effects of impaired methotrexate elimination and of inadvertent overdosages of folic acid antagonists, and to treat megaloblastic anemias due to folic acid deficiency. Also used in combination with 5-fluorouracil to prolong survival in the palliative treatment of patients with advanced colorectal cancer.
Citrovorum factor Pharmacology
Leucovorin is one of several active, chemically reduced derivatives of folic acid. It is useful as an antidote to drugs which act as folic acid antagonists. Leucovorin is a mixture of the diastereoisomers of the 5-formyl derivative of tetrahydrofolic acid (THF). The biologically active compound of the mixture is the (-)-l-isomer, known as Citrovorum factor or (-)-folinic acid. Leucovorin does not require reduction by the enzyme dihydrofolate reductase in order to participate in reactions utilizing folates as a source of “one-carbon” moieties. Administration of leucovorin can counteract the therapeutic and toxic effects of folic acid antagonists such as methotrexate, which act by inhibiting dihydrofolate reductase. Leucovorin has also been used to enhance the activity of fluorouracil.
Citrovorum factor Absorption
Following oral administration, leucovorin is rapidly absorbed. The apparent bioavailability of leucovorin was 97% for 25 mg, 75% for 50 mg, and 37% for 100 mg.
Citrovorum factor side effects and Toxicity
LD50>8000 mg/kg (orally in rats). Excessive amounts of leucovorin may nullify the chemotherapeutic effect of folic acid antagonists.
Citrovorum factor Patient Information
No information avaliable
Citrovorum factor Organisms Affected
Humans and other mammals