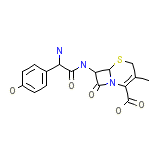
Cefradroxil




Cefradroxil Brand names, Cefradroxil Analogs
Cefradroxil Brand Names Mixture
- None
Cefradroxil Chemical_Formula
C16H17N3O5S
Cefradroxil RX_link
http://www.rxlist.com/cgi/generic/cefadrox.htm
Cefradroxil fda sheet
Cefradroxil msds (material safety sheet)
Cefradroxil Synthesis Reference
L. B. Crast, Jr., U.S. Pat. 3,489,752 (1969)
Cefradroxil Molecular Weight
363.389 g/mol
Cefradroxil Melting Point
197oC
Cefradroxil H2O Solubility
1110 mg/L
Cefradroxil State
Solid
Cefradroxil LogP
-0.355
Cefradroxil Dosage Forms
Capsule; Tablet; Oral liquid
Cefradroxil Indication
For the treatment of the following infections (skin, UTI, ENT) caused by; S. pneumoniae, H. influenzae, staphylococci, S. pyogenes (group A beta-hemolytic streptococci), E. coli, P. mirabilis, Klebsiella sp, coagulase-negative staphylococci and Streptococcus pyogenes
Cefradroxil Pharmacology
Cefadroxil, a first-generation cephalosporin antibiotic, is used to treat urinary tract infections, skin and skin structure infections, pharyngitis, and tonsillitis.
Cefradroxil Absorption
Cefadroxil is well absorbed on oral administration; food does not interfere with its absorption.
Cefradroxil side effects and Toxicity
Nausea, vomiting, diarrhoea, allergic rashes may occur
Cefradroxil Patient Information
Patients should be counseled that intibacterial drugs including DURICEF should only be used to treat bacterial infections. They do not treat viral infections (eg. the common cold). When DURICEF is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelyhood that bacteria will develop resistance and will not be treatable by DURICEF or other antibacterial drugs in the future.
Cefradroxil Organisms Affected
Enteric bacteria and other eubacteria