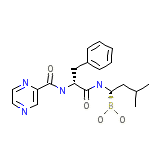
Bortezomib




Bortezomib Brand names, Bortezomib Analogs
Bortezomib Brand Names Mixture
- No information avaliable
Bortezomib Chemical_Formula
C19H25BN4O4
Bortezomib RX_link
http://www.rxlist.com/cgi/generic3/velcade.htm
Bortezomib fda sheet
Bortezomib msds (material safety sheet)
Bortezomib Synthesis Reference
No information avaliable
Bortezomib Molecular Weight
384.237 g/mol
Bortezomib Melting Point
No information avaliable
Bortezomib H2O Solubility
The solubility of bortezomib, as the monomeric boronic acid, is 3.3 to 3.8 mg/mL in a pH range of 2 to 6.5.
Bortezomib State
Solid
Bortezomib LogP
No information avaliable
Bortezomib Dosage Forms
Powder for intravenous injection solution (each single dose vial contains 3.5 mg of bortezomib)
Bortezomib Indication
For treatment of multiple myeloma in patients who have not been successfully treated with at least two previous therapies.
Bortezomib Pharmacology
Bortezomib is a drug that inhibits the mammalian 26S proteasome. The ubiquitin-proteasome pathway plays an essential role in regulating the intracellular concentration of specific proteins, thereby maintaining homeostasis within cells. Inhibition of the 26S proteasome prevents this targeted proteolysis, which can affect multiple signaling cascades within the cell. This disruption of normal homeostatic mechanisms can lead to cell death. Experiments have demonstrated that bortezomib is cytotoxic to a variety of cancer cell types in vitro. Bortezomib causes a delay in tumor growth in vivo in nonclinical tumor models, including multiple myeloma. Tumor cells, that is, rapidly dividing cells, appear to be more sensitive to proteasome inhibition.
Bortezomib Absorption
No information avaliable
Bortezomib side effects and Toxicity
Cardiovascular safety pharmacology studies in monkeys show that lethal IV doses are associated with decreases in blood pressure, increases in heart rate, increases in contractility, and ultimately terminal hypotension. In monkeys, doses of 3.0 mg/m2 and greater (approximately twice the recommended clinical dose) resulted in progressive hypotension starting at 1 hour and progressing to death by 12 to 14 hours following drug administration.
Bortezomib Patient Information
No information avaliable
Bortezomib Organisms Affected
Humans and other mammals