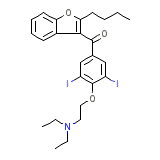
Amiodarone Base




Amiodarone Base Brand names, Amiodarone Base Analogs
Amiodarone Base Brand Names Mixture
- No information avaliable
Amiodarone Base Chemical_Formula
C25H29I2NO3
Amiodarone Base RX_link
http://www.rxlist.com/cgi/generic/amiodarone.htm
Amiodarone Base fda sheet
Amiodarone Base msds (material safety sheet)
Amiodarone Base Synthesis Reference
No information avaliable
Amiodarone Base Molecular Weight
645.312 g/mol
Amiodarone Base Melting Point
No information avaliable
Amiodarone Base H2O Solubility
Low
Amiodarone Base State
Solid
Amiodarone Base LogP
7.706
Amiodarone Base Dosage Forms
Liquid; Solution; Tablet (200 mg and 400 mg)
Amiodarone Base Indication
Intravenously, for initiation of treatment and prophylaxis of frequently recurring ventricular fibrillation and hemodynamically unstable ventricular tachycardia in patients refractory to other therapy. Orally, for the treatment of life-threatening recurrent ventricular arrhythmias such as recurrent ventricular fibrillation and recurrent hemodynamically unstable ventricular tachycardia.
Amiodarone Base Pharmacology
Amiodarone belongs to a class of drugs called Vaughan-Williams Class III antiarrhythmic agents. It is used in the treatment of a wide range of cardiac tachyarhthmias, including both ventricular and supraventricular (atrial) arrhythmias. After intravenous administration in man, amiodarone relaxes vascular smooth muscle, reduces peripheral vascular resistance (afterload), and slightly increases cardiac index. Amiodarone prolongs phase 3 of the cardiac action potential. It has numerous other effects however, including actions that are similar to those of antiarrhythmic classes Ia, II, and IV. Amiodarone shows beta blocker-like and calcium channel blocker-like actions on the SA and AV nodes, increases the refractory period via sodium- and potassium-channel effects, and slows intra-cardiac conduction of the cardiac action potential, via sodium-channel effects.
Amiodarone Base Absorption
Slow and variable (about 20 to 55% of an oral dose is absorbed).
Amiodarone Base side effects and Toxicity
Intravenous, mouse: LD50 = 178 mg/kg. Some side effects have a significant mortality rate: specifically, hepatitis, exacerbation of asthma and congestive failure, and pneumonitis.
Amiodarone Base Patient Information
No information avaliable
Amiodarone Base Organisms Affected
Humans and other mammals