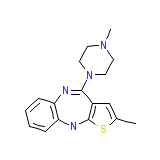
Zyprexa Intramuscular




Categorie
Zyprexa Intramuscular Les marques, Zyprexa Intramuscular Analogs
Zyprexa Intramuscular Les marques melange
Zyprexa Intramuscular Formule chimique
C17H20N4S
Zyprexa Intramuscular RX lien
http://www.rxlist.com/cgi/generic3/symbyax.htm
Zyprexa Intramuscular FDA fiche
Zyprexa Intramuscular msds (fiche de securite des materiaux)
Zyprexa Intramuscular Synthese de reference
JK Chakrabarti et al., US. 5,229,382 (1991)
Zyprexa Intramuscular Poids moleculaire
312.434 g/mol
Zyprexa Intramuscular Point de fusion
195oC
Zyprexa Intramuscular H2O Solubilite
Aucune information disponible
Zyprexa Intramuscular Etat
Solid
Zyprexa Intramuscular LogP
2.199
Zyprexa Intramuscular Formes pharmaceutiques
Tablet (oral)
Zyprexa Intramuscular Indication
Pour le traitement de la schizophrénie et la maniaco-dépression (trouble bipolaire).
Zyprexa Intramuscular Pharmacologie
L'olanzapine, un antipsychotique atypique, est utilisé pour traiter les symptômes positifs et négatifs de la schizophrénie, la manie aiguë avec le trouble bipolaire, l'agitation et des symptômes psychotiques dans la démence. Futur utilisations peuvent inclure le traitement du trouble obsessionnel-compulsif et troubles graves du comportement dans l'autisme. Structurellement et pharmacologique similaire à la clozapine, l'olanzapine se lie à l'alpha (1), la dopamine, , L'histamine H1, muscariniques, et 2 de type sérotonine (5-HT2) récepteurs.
Zyprexa Intramuscular Absorption
Eh bien absorbé, avec environ 40% de la dose métabolisée avant d'atteindre la circulation systémique.
Zyprexa Intramuscular Toxicite
Aucune information disponible
Zyprexa Intramuscular Information pour les patients
PATIENT INFORMATION
Physicians are advised to discuss the following issues with patients for whom they prescribe
olanzapine:
Orthostatic Hypotension: Patients should be advised of the risk of orthostatic hypotension,
especially during the period of initial dose titration and in association with the use of
concomitant drugs that may potentiate the orthostatic effect of olanzapine, e.g., diazepam
or alcohol.
Interference with Cognitive and Motor Performance: Because olanzapine has the potential to
impair judgment, thinking, or motor skills, patients should be cautioned about operating
hazardous machinery, including automobiles, until they are reasonably certain that olanzapine
therapy does not affect them adversely.
Pregnancy: Patients should be advised to notify their physician if they become pregnant or
intend to become pregnant during therapy with olanzapine.
Nursing: Patients should be advised not to breast-feed an infant if they are taking olanzapine.
Concomitant Medication: Patients should be advised to inform their physicians if they are taking,
or plan to take, any prescription or over-the-counter drugs, since there is a potential for
interactions.
Alcohol: Patients should be advised to avoid alcohol while taking olanzapine.
Heat Exposure and Dehydration: Patients should be advised regarding appropriate care in avoiding
overheating and dehydration.
Phenylketonurics: ZYPREXA ZYDIS (olanzapine orally disintegrating tablets) contains phenylalanine
(0.34, 0.45, 0.67, or 0.90 mg per 5, 10, 15, or 20mg tablet, respectively).
Laboratory Tests
Periodic assessment of transaminases is recommended in patients with significant hepatic disease.
Physicians are advised to discuss the following issues with patients for whom they prescribe
olanzapine:
Orthostatic Hypotension: Patients should be advised of the risk of orthostatic hypotension,
especially during the period of initial dose titration and in association with the use of
concomitant drugs that may potentiate the orthostatic effect of olanzapine, e.g., diazepam
or alcohol.
Interference with Cognitive and Motor Performance: Because olanzapine has the potential to
impair judgment, thinking, or motor skills, patients should be cautioned about operating
hazardous machinery, including automobiles, until they are reasonably certain that olanzapine
therapy does not affect them adversely.
Pregnancy: Patients should be advised to notify their physician if they become pregnant or
intend to become pregnant during therapy with olanzapine.
Nursing: Patients should be advised not to breast-feed an infant if they are taking olanzapine.
Concomitant Medication: Patients should be advised to inform their physicians if they are taking,
or plan to take, any prescription or over-the-counter drugs, since there is a potential for
interactions.
Alcohol: Patients should be advised to avoid alcohol while taking olanzapine.
Heat Exposure and Dehydration: Patients should be advised regarding appropriate care in avoiding
overheating and dehydration.
Phenylketonurics: ZYPREXA ZYDIS (olanzapine orally disintegrating tablets) contains phenylalanine
(0.34, 0.45, 0.67, or 0.90 mg per 5, 10, 15, or 20mg tablet, respectively).
Laboratory Tests
Periodic assessment of transaminases is recommended in patients with significant hepatic disease.
Zyprexa Intramuscular Organismes affectes
Les humains et autres mammifères