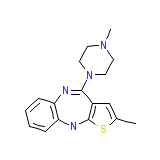
Olansek




Olansek Brand names, Olansek Analogs
Olansek Brand Names Mixture
- No information avaliable
Olansek Chemical_Formula
C17H20N4S
Olansek RX_link
http://www.rxlist.com/cgi/generic3/symbyax.htm
Olansek fda sheet
Olansek msds (material safety sheet)
Olansek Synthesis Reference
J. K. Chakrabarti et al., U.S. Pat. 5,229,382 (1991)
Olansek Molecular Weight
312.434 g/mol
Olansek Melting Point
195oC
Olansek H2O Solubility
No information avaliable
Olansek State
Solid
Olansek LogP
2.199
Olansek Dosage Forms
Tablet (oral)
Olansek Indication
For the treatment of schizophrenia and manic depression (bipolar disorder).
Olansek Pharmacology
Olanzapine, an atypical antipsychotic agent, is used to treat both negative and positive symptoms of schizophrenia, acute mania with bipolar disorder, agitation, and psychotic symptoms in dementia. Future uses may include the treatment of obsessive-compulsive disorder and severe behavioral disorders in autism. Structurally and pharmacologically similar to clozapine, olanzapine binds to alpha(1), dopamine, histamine H1, muscarinic, and serotonin type 2 (5-HT2) receptors.
Olansek Absorption
Well absorbed, with approximately 40% of the dose metabolized before reaching the systemic circulation.
Olansek side effects and Toxicity
No information avaliable
Olansek Patient Information
PATIENT INFORMATION
Physicians are advised to discuss the following issues with patients for whom they prescribe
olanzapine:
Orthostatic Hypotension: Patients should be advised of the risk of orthostatic hypotension,
especially during the period of initial dose titration and in association with the use of
concomitant drugs that may potentiate the orthostatic effect of olanzapine, e.g., diazepam
or alcohol.
Interference with Cognitive and Motor Performance: Because olanzapine has the potential to
impair judgment, thinking, or motor skills, patients should be cautioned about operating
hazardous machinery, including automobiles, until they are reasonably certain that olanzapine
therapy does not affect them adversely.
Pregnancy: Patients should be advised to notify their physician if they become pregnant or
intend to become pregnant during therapy with olanzapine.
Nursing: Patients should be advised not to breast-feed an infant if they are taking olanzapine.
Concomitant Medication: Patients should be advised to inform their physicians if they are taking,
or plan to take, any prescription or over-the-counter drugs, since there is a potential for
interactions.
Alcohol: Patients should be advised to avoid alcohol while taking olanzapine.
Heat Exposure and Dehydration: Patients should be advised regarding appropriate care in avoiding
overheating and dehydration.
Phenylketonurics: ZYPREXA ZYDIS (olanzapine orally disintegrating tablets) contains phenylalanine
(0.34, 0.45, 0.67, or 0.90 mg per 5, 10, 15, or 20mg tablet, respectively).
Laboratory Tests
Periodic assessment of transaminases is recommended in patients with significant hepatic disease.
Physicians are advised to discuss the following issues with patients for whom they prescribe
olanzapine:
Orthostatic Hypotension: Patients should be advised of the risk of orthostatic hypotension,
especially during the period of initial dose titration and in association with the use of
concomitant drugs that may potentiate the orthostatic effect of olanzapine, e.g., diazepam
or alcohol.
Interference with Cognitive and Motor Performance: Because olanzapine has the potential to
impair judgment, thinking, or motor skills, patients should be cautioned about operating
hazardous machinery, including automobiles, until they are reasonably certain that olanzapine
therapy does not affect them adversely.
Pregnancy: Patients should be advised to notify their physician if they become pregnant or
intend to become pregnant during therapy with olanzapine.
Nursing: Patients should be advised not to breast-feed an infant if they are taking olanzapine.
Concomitant Medication: Patients should be advised to inform their physicians if they are taking,
or plan to take, any prescription or over-the-counter drugs, since there is a potential for
interactions.
Alcohol: Patients should be advised to avoid alcohol while taking olanzapine.
Heat Exposure and Dehydration: Patients should be advised regarding appropriate care in avoiding
overheating and dehydration.
Phenylketonurics: ZYPREXA ZYDIS (olanzapine orally disintegrating tablets) contains phenylalanine
(0.34, 0.45, 0.67, or 0.90 mg per 5, 10, 15, or 20mg tablet, respectively).
Laboratory Tests
Periodic assessment of transaminases is recommended in patients with significant hepatic disease.
Olansek Organisms Affected
Humans and other mammals