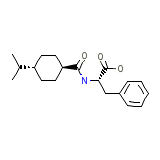
Starsis




Starsis Brand names, Starsis Analogs
Starsis Brand Names Mixture
- No information avaliable
Starsis Chemical_Formula
C19H27NO3
Starsis RX_link
http://www.rxlist.com/cgi/generic2/nateglinide.htm
Starsis fda sheet
Starsis msds (material safety sheet)
Starsis Synthesis Reference
No information avaliable
Starsis Molecular Weight
317.423 g/mol
Starsis Melting Point
No information avaliable
Starsis H2O Solubility
Practically insoluble
Starsis State
Solid
Starsis LogP
3.824
Starsis Dosage Forms
Tablets (60, 120 mg)
Starsis Indication
For use as monotherapy to lower blood glucose in patients with Type 2 diabetes (non-insulin dependent diabetes mellitus, NIDDM) whose hyperglycemia cannot be adequately controlled by diet and physical exercise and who have not been chronically treated with other anti-diabetic agents. Also indicated for use in combination with metformin.
Starsis Pharmacology
Nateglinide is an amino-acid derivative that lowers blood glucose levels by stimulating insulin secretion from the pancreas. This action is dependent upon functioning beta-cells in the pancreatic islets.
Starsis Absorption
Rapidly absorbed following oral administration prior to a meal, absolute bioavailability is estimated to be approximately 73%.
Starsis side effects and Toxicity
An overdose may result in an exaggerated glucose-lowering effect with the development of hypoglycemic symptoms.
Starsis Patient Information
Starsis Organisms Affected
Humans and other mammals