Leustatin




Leustatin Brand names, Leustatin Analogs
Leustatin Brand Names Mixture
- No information avaliable
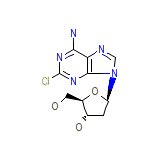
Leustatin Chemical_Formula
C10H12ClN5O3
Leustatin RX_link
http://www.rxlist.com/cgi/generic3/cladribine.htm
Leustatin fda sheet
Leustatin msds (material safety sheet)
Leustatin Synthesis Reference
No information avaliable
Leustatin Molecular Weight
285.687 g/mol
Leustatin Melting Point
215 oC
Leustatin H2O Solubility
No information avaliable
Leustatin State
Solid
Leustatin LogP
1.57
Leustatin Dosage Forms
Solution
Leustatin Indication
For the treatment of active hairy cell leukemia (leukemic reticuloendotheliosis) as defined by clinically significant anemia, neutropenia, thrombocytopenia, or disease-related symptoms.
Leustatin Pharmacology
Cladribine is a synthetic antineoplastic agent with immunosuppressive effects. Cladribine is one of a group of chemotherapy drugs known as the anti-metabolites. Anti-metabolites stop cells making and repairing DNA. Cancer cells need to make and repair DNA in order to grow and multiply.
Leustatin Absorption
Oral bioavailability is 34 to 48%.
Leustatin side effects and Toxicity
Symptoms of overdose include irreversible neurologic toxicity (paraparesis/quadriparesis), acute nephrotoxicity, and severe bone marrow suppression resulting in neutropenia, anemia and thrombocytopenia.
Leustatin Patient Information
LEUSTATIN (cladribine) Injection should be administered under the supervision of a qualified physician experienced in the use of antineoplastic therapy. Suppression of bone marrow function should be anticipated. This is usually reversible and appears to be dose dependent. Serious neurological toxicity (including irreversible paraparesis and quadraparesis) has been reported in patients who received LEUSTATIN Injection by continuous infusion at high doses (4 to 9 times the recommended dose for Hairy Cell Leukemia). Neurologic toxicity appears to demonstrate a dose relationship; however, severe neurological toxicity has been reported rarely following treatment with standard cladribine dosing regimens. Acute nephrotoxicity has been observed with high doses of LEUSTATIN (4 to 9 times the recommended dose for Hairy Cell Leukemia), especially when given concomitantly with other nephrotoxic agents/ therapies.
Leustatin Organisms Affected
Humans and other mammals