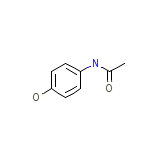
Finimal




Finimal Brand names, Finimal Analogs
- APAP
- Abenol
- Abensanil
- Acamol
- Accu-Tap
- Acephen
- Aceta Elixir
- Aceta Tablets
- Acetagesic
- Acetalgin
- Acetaminofen
- Actamin
- Actimol
- Algotropyl
- Allay
- Alpiny
- Alpinyl
- Alvedon
- Amadil
- Aminofen
- Anacin
- Anacin-3
- Anaflon
- Anapap
- Anelix
- Anexsia
- Anhiba
- Anoquan
- Apacet
- Apadon
- Apamid
- Apamide
- Apo-Acetaminophen
- Atasol
- Bancap
- Banesin
- Bayer Select
- Bickie-mol
- Bucet
- Butapap
- Calpol
- Captin
- Cetadol
- Clixodyne
- Co-Gesic
- Conacetol
- Dafalgan
- Dapa
- Dapa X-S
- Darvocet
- Datril
- DHC Plus
- Dimindol
- Dirox
- Disprol
- Dolene AP-65
- Doliprane
- Dolprone
- Drixoral Plus
- Dularin
- Duradyne DHC
- Dymadon
- Dypap
- Elixodyne
- Enelfa
- Eneril
- Esgic
- Esgic-Plus
- Eu-Med
- Excedrin
- Exdol
- Febridol
- Febrilix
- Febrinol
- Febro-Gesic
- Febrolin
- Femcet
- Fendon
- Feverall
- Fevor
- Finimal
- Fioricet
- Gelocatil
- Genapap
- Genebs
- Hedex
- Homoolan
- Hy-Phen
- Hydrocet
- Injectapap
- Janupap
- Korum
- Lestemp
- Liquagesic
- Liquiprin
- Lonarid
- Lorcet-Hd
- Lortab
- Lyteca
- Medigesic Plus
- Momentum
- Multin
- NAPA
- Napafen
- Napap
- Naprinol
- Nealgyl
- Nebs
- Neopap
- Neotrend
- Nobedon
- Norcet
- Norco
- Oraphen-PD
- Ortensan
- Oxycet
- Pacemo
- Painex
- Paldesic
- Panadol
- Panaleve
- Panasorb
- Panets
- Panex
- Panofen
- Papa-Deine
- Paracet
- Paracetamol
- Paracetamolo
- Paracetanol
- Parapan
- Paraspen
- Parelan
- Parmol
- Pasolind
- Pasolind N
- Pedric
- Percocet
- Phenaphen
- Phenaphen Caplets
- Phendon
- Phrenilin
- Phrenilin Forte
- Prompt
- Propacet 100
- Proval #3
- Pyrinazine
- Redutemp
- Rivalgyl
- Robigesic
- Rounox
- Roxicet
- Roxilox
- SK-Apap
- Salzone
- Sedapap
- Servigesic
- Snaplets-FR
- St. Joseph Fever Reducer
- Suppap
- Synalgos-Dc-A
- Tabalgin
- Talacen
- Tapanol
- Tapar
- Tavist Allergy/Sinus/Headache
- Temlo
- Tempanal
- Tempra
- Tencon
- Tibinide
- Tibizide
- Tisin
- Tisiodrazida
- Tizide
- Tralgon
- Triad
- Triaprin
- Tussapap
- Tycolet
- Tylenol
- Tylox
- Tylox-325
- Ultracet
- Valadol
- Valgesic
- Valorin
- Valorin Extra
- Vicodin
- Vicodin Es
- Vicodin Hp
- Wygesic
Finimal Brand Names Mixture
- Amacodone (acetaminophen + hydrocodone bitartrate)
- Anexsia (acetaminophen + hydrocodone bitartrate)
- Anodynos (acetaminophen + hydrocodone bitartrate)
- Anolor (acetaminophen + butalbital + caffeine)
- Bancap (acetaminophen + hydrocodone bitartrate)
- CoGesic (acetaminophen + hydrocodone bitartrate)
- Dolacet (acetaminophen + hydrocodone bitartrate)
- Duradyne (acetaminophen + hydrocodone bitartrate)
- Endolor (acetaminophen + butalbital + caffeine)
- Esgic (acetaminophen + butalbital + caffeine)
- Fioricet (acetaminophen + butalbital + caffeine)
- Hydrocet (acetaminophen + hydrocodone bitartrate)
- Hydrogesic (acetaminophen + hydrocodone bitartrate)
- Margesic (acetaminophen + hydrocodone bitartrate)
- Lorcet (acetaminophen + hydrocodone bitartrate)
- Lortab (acetaminophen + hydrocodone bitartrate)
- Norcet (acetaminophen + hydrocodone bitartrate)
- Oxycet (acetaminophen + oxycodone hydrochloride)
- Percocet (acetaminophen + oxycodone hydrochloride)
- Roxicet (acetaminophen + oxycodone hydrochloride)
- Roxilox (acetaminophen + oxycodone hydrochloride)
- Stagesic (acetaminophen + hydrocodone bitartrate)
- TGesic (acetaminophen + hydrocodone bitartrate)
- Tylox (acetaminophen + oxycodone hydrochloride)
- Vicodin (acetaminophen + hydrocodone bitartrate)
- Zebutal (acetaminophen + butalbital + caffeine)
- Zydone (acetaminophen + hydrocodone bitartrate)
Finimal Chemical_Formula
C8H9NO2
Finimal RX_link
http://www.rxlist.com/cgi/generic/apap.htm
Finimal fda sheet
Finimal msds (material safety sheet)
Finimal Synthesis Reference
Pearson et al., J. Am .Chem. Soc. 75, 5907 (1953)
Finimal Molecular Weight
151.163 g/mol
Finimal Melting Point
169-170.5oC
Finimal H2O Solubility
14 mg/mL (very slightly soluble in cold water; considerably more soluble in hot water)
Finimal State
Solid
Finimal LogP
0.917
Finimal Dosage Forms
Capsule; Drops; Elixir; Liquid; Solution; Suspension; Syrup; Tablet; Tablet (effervescent)
Finimal Indication
For temporary relief of fever and minor aches and pains.
Finimal Pharmacology
Acetaminophen (USAN) or Paracetamol (INN) is a popular analgesic and antipyretic drug that is used for the relief of fever, headaches, and other minor aches and pains. It is a major ingredient in numerous cold and flu medications and many prescription analgesics. It is extremely safe in standard doses, but because of its wide availability, deliberate or accidental overdoses are not uncommon. Acetaminophen, unlike other common analgesics such as aspirin and ibuprofen, has no anti-inflammatory properties or effects on platelet function, and so it is not a member of the class of drugs known as non-steroidal anti-inflammatory drugs or NSAIDs. In normal doses acetaminophen does not irritate the lining of the stomach nor affect blood coagulation, the kidneys, or the fetal ductus arteriosus (as NSAIDs can). Like NSAIDs and unlike opioid analgesics, acetaminophen does not cause euphoria or alter mood in any way. Acetaminophen and NSAIDs have the benefit of being completely free of problems with addiction, dependence, tolerance and withdrawal. Acetaminophen is used on its own or in combination with pseudoephedrine, dextromethorphan, chlorpheniramine, diphenhydramine, doxylamine, codeine, hydrocodone, or oxycodone.
Finimal Absorption
Rapid and almost complete
Finimal side effects and Toxicity
Oral, mouse: LD50 = 338 mg/kg; Oral, rat: LD50 = 1944 mg/kg. Acetaminophen is metabolized primarily in the liver, where most of it is converted to inactive compounds by conjugation with sulfate and glucuronide, and then excreted by the kidneys. Only a small portion is metabolized via the hepatic cytochrome P450 enzyme system. The toxic effects of acetaminophen are due to a minor alkylating metabolite (N-acetyl-p-benzo-quinone imine), not acetaminophen itself nor any of the major metabolites. This toxic metabolite reacts with sulfhydryl groups. At usual doses, it is quickly detoxified by combining irreversibly with the sulfhydryl group of glutathione to produce a non-toxic conjugate that is eventually excreted by the kidneys. The toxic dose of paracetamol is highly variable. In adults, single doses above 10 grams or 140 mg/kg have a reasonable likelihood of causing toxicity. In adults, single doses of more than 25 grams have a high risk of lethality.
Finimal Patient Information
Do not use the maximum dosage of this product for more than 10 days except under the advice
and supervision of a physician. Do not take the product for pain for more than 10 days,
or for fever for more than 3 days unless directed by a physician. If pain or fever persists
or gets worse, if new symptoms occur, or if redness or swelling is present, consult a
physician because these could be signs of a serious condition. Do not use with other
products containing acetaminophen.
Do not use if carton is opened. Keep this and all medication out of The reach of children.
As with any drug, if you are pregnant or nursing A baby, seek the advice of a health
professional before using this product. In the case of accidental overdose, contact a
doctor or a poison control Center immediately. Prompt medical attention is critical for
adults as Well as for children even if you do not notice any signs or symptoms.
and supervision of a physician. Do not take the product for pain for more than 10 days,
or for fever for more than 3 days unless directed by a physician. If pain or fever persists
or gets worse, if new symptoms occur, or if redness or swelling is present, consult a
physician because these could be signs of a serious condition. Do not use with other
products containing acetaminophen.
Do not use if carton is opened. Keep this and all medication out of The reach of children.
As with any drug, if you are pregnant or nursing A baby, seek the advice of a health
professional before using this product. In the case of accidental overdose, contact a
doctor or a poison control Center immediately. Prompt medical attention is critical for
adults as Well as for children even if you do not notice any signs or symptoms.
Finimal Organisms Affected
Humans and other mammals