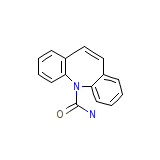
Equetro




Equetro Brand names, Equetro Analogs
- Apo-Carbamazepine
- Atretol
- Biston
- Calepsin
- Carbamazepen
- Carbamezepine
- Carbatrol
- Carbazepine
- Carbelan
- Epitol
- Equetro
- Finlepsin
- Karbamazepin
- Lexin
- Neurotol
- Novo-Carbamaz
- Nu-Carbamazepine
- Sirtal
- Stazepin
- Stazepine
- Taro-Carbamazepine
- Taro-Carbamazepine Cr
- Tegretal
- Tegretol
- Tegretol Chewtabs
- Tegretol Cr
- Tegretol-Xr
- Telesmin
- Teril
- Timonil
Equetro Brand Names Mixture
- No information avaliable
Equetro Chemical_Formula
C15H12N2O
Equetro RX_link
http://www.rxlist.com/cgi/generic/carbam.htm
Equetro fda sheet
Equetro msds (material safety sheet)
Equetro Synthesis Reference
Schindler, U.S. Pat. 2,948,718 (1960)
Equetro Molecular Weight
236.269 g/mol
Equetro Melting Point
190.2 oC
Equetro H2O Solubility
17.7 mg/L
Equetro State
Solid
Equetro LogP
2.69
Equetro Dosage Forms
Suspension; Tablet; Tablet (extended-release)
Equetro Indication
For the treatment of epilepsy and pain associated with true trigeminal neuralgia.
Equetro Pharmacology
Carbamazepine, an anticonvulsant structurally similar to tricyclic antidepressants, is used to treat partial seizures, tonic-clonic seizures, pain of neurologic origin such as trigeminal neuralgia, and psychiatric disorders including manic-depressive illness and aggression due to dementia.
Equetro Absorption
No information avaliable
Equetro side effects and Toxicity
Mild ingestions cause vomiting, drowsiness, ataxia, slurred speech, nystagmus, dystonic reactions, and hallucinations. Severe intoxications may produce coma, seizures, respiratory depression, and hypotension
Equetro Patient Information
PATIENT INFORMATION
Carbamazepine is used for the treatment of seizures. It also is used to treat certain types of nerve pain. Inform your physican if you are pregnant or nursing. Inform your physician if you have glaucoma. Do not take this medication with a monoamine oxidase inhibitor. This medication may cause dizziness, drowsiness, or blurred vision; use caution while driving or operating hazardous machinery. Do not stop taking carbamazepine without talking with your physician. Shake the suspension well before each use. This medication should be taken with meals to avoid stomach upset. Notify your physician if you develop unexplained fever, sore throat, unusual bleeding or bruising, or yellow eyes or skin.
Carbamazepine is used for the treatment of seizures. It also is used to treat certain types of nerve pain. Inform your physican if you are pregnant or nursing. Inform your physician if you have glaucoma. Do not take this medication with a monoamine oxidase inhibitor. This medication may cause dizziness, drowsiness, or blurred vision; use caution while driving or operating hazardous machinery. Do not stop taking carbamazepine without talking with your physician. Shake the suspension well before each use. This medication should be taken with meals to avoid stomach upset. Notify your physician if you develop unexplained fever, sore throat, unusual bleeding or bruising, or yellow eyes or skin.
Equetro Organisms Affected
Humans and other mammals