Cefzon (TN)




Cefzon (TN) Brand names, Cefzon (TN) Analogs
Cefzon (TN) Brand Names Mixture
- No information avaliable
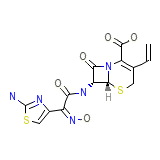
Cefzon (TN) Chemical_Formula
C14H13N5O5S2
Cefzon (TN) RX_link
http://www.rxlist.com/cgi/generic2/cefdin.htm
Cefzon (TN) fda sheet
Cefzon (TN) msds (material safety sheet)
Cefzon (TN) Synthesis Reference
No information avaliable
Cefzon (TN) Molecular Weight
395.416 g/mol
Cefzon (TN) Melting Point
No information avaliable
Cefzon (TN) H2O Solubility
No information avaliable
Cefzon (TN) State
Solid
Cefzon (TN) LogP
0.657
Cefzon (TN) Dosage Forms
Capsules for oral administration (300mg); Oral suspension (125mg per 5mL solution)
Cefzon (TN) Indication
For the treatment of the respiratory, skin, soft tissue, and ENT infections caused by H. influenzae (including b-lactamase producing strains), H. parainfluenzae (including b-lactamase producing strains), S. pneumoniae (penicillin-susceptible strains), S. pyogenes, S. aureus (including b-lactamase producing strains), and M. catarrhalis.
Cefzon (TN) Pharmacology
Cefdinir is a third generation cephalosporin with a broad spectrum of activity against enteric gram-negative rods. Cefdinir is stable in the presence of some, but not all, b-lactamase enzymes. As a result, many organisms resistant to penicillins and some cephalosporins are susceptible to cefdinir. Cephalosporins work the same way as penicillins: they interfere with the peptidoglycan synthesis of the bacterial wall by inhibiting the final transpeptidation needed for the cross-links. This effect is bactericidal.
Cefzon (TN) Absorption
Maximal plasma cefdinir concentrations occur 2 to 4 hours postdose following capsule or suspension administration. Estimated bioavailability of cefdinir capsules is 21% following administration of a 300 mg capsule dose, and 16% following administration of a 600 mg capsule dose. Estimated absolute bioavailability of cefdinir suspension is 25%. Absorption is reduced by approximately 15% when administered with a high fat meal.
Cefzon (TN) side effects and Toxicity
Information on cefdinir overdosage in humans is not available. In acute rodent toxicity studies, a single oral 5600-mg/kg dose produced no adverse effects. Toxic signs and symptoms following overdosage with other b-lactam antibiotics have included nausea, vomiting, epigastric distress, diarrhea, and convulsions.
Cefzon (TN) Patient Information
Cefzon (TN) Organisms Affected
Enteric gram-negative rods