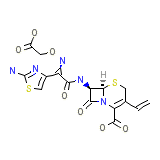
Cefixime Anhydrous




Cefixime Anhydrous Brand names, Cefixime Anhydrous Analogs
Cefixime Anhydrous Brand Names Mixture
- No information avaliable
Cefixime Anhydrous Chemical_Formula
C16H15N5O7S2
Cefixime Anhydrous RX_link
http://www.rxlist.com/cgi/generic/cefixime.htm
Cefixime Anhydrous fda sheet
Cefixime Anhydrous msds (material safety sheet)
Cefixime Anhydrous Synthesis Reference
T. Takaya et al., U.S. Pat. 4,409,214 (1981)
Cefixime Anhydrous Molecular Weight
453.452 g/mol
Cefixime Anhydrous Melting Point
218-225 oC
Cefixime Anhydrous H2O Solubility
55.11 mg/L
Cefixime Anhydrous State
Solid
Cefixime Anhydrous LogP
-0.559
Cefixime Anhydrous Dosage Forms
Oral suspension (100 mg/5 mL); Tablet (200 mg and 400 mg); Powder for solution; Powder for suspension
Cefixime Anhydrous Indication
For use in the treatment of the following infections when caused by susceptible strains of the designated microorganisms: (1) uncomplicated urinary tract infections caused by Escherichia coli and Proteus mirabilis, (2) otitis media caused by Haemophilus influenzae (beta-lactamase positive and negative strains), Moraxella catarrhalis (most of which are beta-lactamase positive), and S. pyogenes, (3) pharyngitis and tonsillitis caused by S. pyogenes, (4) acute bronchitis and acute exacerbations of chronic bronchitis caused by Streptococcus pneumoniae and Haemophilus influenzae (beta-lactamase positive and negative strains), and (5) uncomplicated gonorrhea (cervical/urethral) caused by Neisseria gonorrhoeae (penicillinase- and non-penicillinase-producing strains).
Cefixime Anhydrous Pharmacology
Cefixime, an antibiotic, is a third-generation cephalosporin like ceftriaxone and cefotaxime. Cefixime is highly stable in the presence of beta-lactamase enzymes. As a result, many organisms resistant to penicillins and some cephalosporins due to the presence of beta-lactamases, may be susceptible to cefixime. The antibacterial effect of cefixime results from inhibition of mucopeptide synthesis in the bacterial cell wall.
Cefixime Anhydrous Absorption
About 40%-50% absorbed orally whether administered with or without food, however, time to maximal absorption is increased approximately 0.8 hours when administered with food.
Cefixime Anhydrous side effects and Toxicity
Symptoms of overdose include blood in the urine, diarrhea, nausea, upper abdominal pain, and vomiting.
Cefixime Anhydrous Patient Information
Cefixime Anhydrous Organisms Affected
Enteric bacteria and other eubacteria