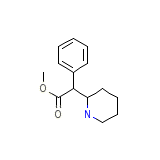
Riphenidate




Riphenidate Brand names, Riphenidate Analogs
- 4311/B Ciba
- Calocain
- Centedein
- Centedrin
- Centedrine
- Centredin
- Concerta
- Focalin
- Focalin XR
- Methylphen
- Meridil
- Metadate
- Metadate CD
- Metadate ER
- Methyl phenidyl acetate
- Methylin
- Methylin ER
- Methylofenidan
- Methylphenidan
- Methylphenidate HCl
- Methylphenidate hydrochloride
- Methylphenidatum [Inn-Latin]
- Methylphenidylacetate hydrochloride
- Methypatch
- Metilfenidat hydrochloride
- Metilfenidato [Inn-Spanish]
- Metilfenidato [Italian]
- Phenidylate
- Plimasine
- PMS-Methylphenidate
- Riphenidate
- Ritalin
- Ritalin hydrochloride
- Ritalin LA
- Ritalin SR
- Ritalin-SR
- Ritaline
- Ritcher Works
Riphenidate Brand Names Mixture
- No information avaliable
Riphenidate Chemical_Formula
C14H19NO2
Riphenidate RX_link
http://www.rxlist.com/cgi/generic/methphen.htm
Riphenidate fda sheet
Riphenidate msds (material safety sheet)
Riphenidate Synthesis Reference
M. Hartmann, L. Panizzon, U.S. Pat. 2,507,631 (1950)
Riphenidate Molecular Weight
233.306 g/mol
Riphenidate Melting Point
224-226oC
Riphenidate H2O Solubility
1255mg/L
Riphenidate State
Solid
Riphenidate LogP
3.192
Riphenidate Dosage Forms
Tablets; Tablets (sustained release)
Riphenidate Indication
For use as an integral part of a total treatment program which typically includes other remedial measures (psychological, educational, social) for a stabilizing effect in children with a behavioral syndrome characterized by the following group of developmentally inappropriate symptoms: moderate-to-severe distractibility, short attention span, hyperactivity, emotional lability, and impulsivity.
Riphenidate Pharmacology
Methylphenidate is a central nervous system stimulant used most commonly in the treatment of attention-deficit disorders in children and for narcolepsy. Its mechanisms appear to be similar to those of dextroamphetamine.
Riphenidate Absorption
Readily absorbed in a biphasic manner. It reaches peak absorption at approximately two hours for the first phase and five hours for the second phase. Bioavailability is low (approximately 30%)
Riphenidate side effects and Toxicity
Symptoms of overdose include vomiting, agitation, tremors, hyperreflexia, muscle twitching, convulsions (may be followed by coma), euphoria, confusion, hallucinations, delirium, sweating, flushing, headache, hyperpyrexia, tachycardia, palpitations, cardiac arrhythmias, hypertension, mydriasis, and dryness of mucous membranes. LD50=190mg/kg (orally in mice)
Riphenidate Patient Information
Riphenidate Organisms Affected
Humans and other mammals