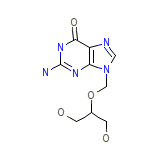
GA2




Categoria
GA2 Nombres de marca, GA2 Analogos
GA2 Marca los nombres de mezcla
GA2 Formula quimica
C9H13N5O4
GA2 RX enlace
http://www.rxlist.com/cgi/generic2/vitrasert.htm
GA2 FDA hoja
GA2 MSDS (hoja de seguridad de materiales)
GA2 Sintesis de referencia
Alhede, Boerge, et al;. J. Org;. 56, 6, 2139 a 2143 (1991)
GA2 Peso molecular
255.231 g/mol
GA2 Punto de fusion
250 oC
GA2 H2O Solubilidad
GA2 Estado
Solid
GA2 LogP
-1.438
GA2 Formas de dosificacion
Oculares insertar, Cápsula oral, por inyección
GA2 Indicacion
Para la inducción y mantenimiento en el tratamiento de la infección por citomegalovirus (CMV) en pacientes inmunocomprometidos, incluyendo pacientes con síndrome de inmunodeficiencia adquirida (SIDA). También se utiliza en el tratamiento
GA2 Farmacologia
El ganciclovir es un nucleósido análogo sintético de la 2'-desoxiguanosina, que inhibe la replicación del virus del herpes tanto in vitro como in vivo. Sensibles a los virus humanos incluyen el citomegalovirus (CMV), herpes simplex virus 1 y 2 (HSV-1, HSV-2), virus de Epstein-Barr (EBV) y el virus varicela-zóster (VVZ), sin embargo los estudios clínicos se han limitado a la evaluación de la eficacia en pacientes con infección por CMV. Ganciclovir la maquinaria viral necesaria para transmitir el virus a otras células.
GA2 Absorcion
GA2 Toxicidad
Oral, DL50:> 2g/kg. Por vía intravenosa, el perro LD50: 150mg/kg>. Los síntomas de sobredosis incluyen pancitopenia irreversible, empeoramiento de los síntomas GI, e insuficiencia renal aguda. Agente de sospecha de cáncer.
GA2 Informacion de Pacientes
All patients should be informed that the major toxicities of ganciclovir are granulocytopenia
(neutropenia), anemia and thrombocytopenia and that dose modifications may be required, including
discontinuation. The importance of close monitoring of blood counts while on therapy should be emphasized.
Patients should be informed that ganciclovir has been associated with elevations in serum creatinine.
Patients should be instructed to take CYTOVENE capsules with food to maximize bioavailability.
Patients should be advised that ganciclovir has caused decreased sperm production in animals and may cause
infertility in humans. Women of childbearing potential should be advised that ganciclovir causes birth
defects in animals and should not be used during pregnancy. Women of childbearing potential should be
advised to use effective contraception during treatment with CYTOVENE-IV or CYTOVENE. Similarly, men
should be advised to practice barrier contraception during and for at least 90 days following treatment
with CYTOVENE-IV or CYTOVENE.
Patients should be advised that ganciclovir causes tumors in animals. Although there is no information
from human studies, ganciclovir should be considered a potential carcinogen.
All HIV+ Patients: These patients may be receiving zidovudine (Retrovir?*). Patients should be counseled
that treatment with both ganciclovir and zidovudine simultaneously may not be tolerated by some patients
and may result in severe granulocytopenia (neutropenia). Patients with AIDS may be receiving didanosine
(Videx?#). Patients should be counseled that concomitant treatment with both ganciclovir and didanosine
can cause didanosine serum concentrations to be significantly increased.
HIV+ Patients With CMV Retinitis: Ganciclovir is not a cure for CMV retinitis, and immunocompromised
patients may continue to experience progression of retinitis during or following treatment. Patients
should be advised to have ophthalmologic follow-up examinations at a minimum of every 4 to 6 weeks while
being treated with CYTOVENE-IV or CYTOVENE. Some patients will require more frequent follow-up.
Transplant Recipients: Transplant recipients should be counseled regarding the high frequency of impaired
renal function in transplant recipients who received CYTOVENE-IV solution in controlled clinical trials,
particularly in patients receiving concomitant administration of nephrotoxic agents such as cyclosporine
and amphotericin B. Although the specific mechanism of this toxicity, which in most cases was reversible,
has not been determined, the higher rate of renal impairment in patients receiving CYTOVENE-IV solution
compared with those who received placebo in the same trials may indicate that CYTOVENE-IV played a
significant role.
(neutropenia), anemia and thrombocytopenia and that dose modifications may be required, including
discontinuation. The importance of close monitoring of blood counts while on therapy should be emphasized.
Patients should be informed that ganciclovir has been associated with elevations in serum creatinine.
Patients should be instructed to take CYTOVENE capsules with food to maximize bioavailability.
Patients should be advised that ganciclovir has caused decreased sperm production in animals and may cause
infertility in humans. Women of childbearing potential should be advised that ganciclovir causes birth
defects in animals and should not be used during pregnancy. Women of childbearing potential should be
advised to use effective contraception during treatment with CYTOVENE-IV or CYTOVENE. Similarly, men
should be advised to practice barrier contraception during and for at least 90 days following treatment
with CYTOVENE-IV or CYTOVENE.
Patients should be advised that ganciclovir causes tumors in animals. Although there is no information
from human studies, ganciclovir should be considered a potential carcinogen.
All HIV+ Patients: These patients may be receiving zidovudine (Retrovir?*). Patients should be counseled
that treatment with both ganciclovir and zidovudine simultaneously may not be tolerated by some patients
and may result in severe granulocytopenia (neutropenia). Patients with AIDS may be receiving didanosine
(Videx?#). Patients should be counseled that concomitant treatment with both ganciclovir and didanosine
can cause didanosine serum concentrations to be significantly increased.
HIV+ Patients With CMV Retinitis: Ganciclovir is not a cure for CMV retinitis, and immunocompromised
patients may continue to experience progression of retinitis during or following treatment. Patients
should be advised to have ophthalmologic follow-up examinations at a minimum of every 4 to 6 weeks while
being treated with CYTOVENE-IV or CYTOVENE. Some patients will require more frequent follow-up.
Transplant Recipients: Transplant recipients should be counseled regarding the high frequency of impaired
renal function in transplant recipients who received CYTOVENE-IV solution in controlled clinical trials,
particularly in patients receiving concomitant administration of nephrotoxic agents such as cyclosporine
and amphotericin B. Although the specific mechanism of this toxicity, which in most cases was reversible,
has not been determined, the higher rate of renal impairment in patients receiving CYTOVENE-IV solution
compared with those who received placebo in the same trials may indicate that CYTOVENE-IV played a
significant role.
GA2 Organismos afectados
El herpes virus humano