Zrivada




Zrivada Brand names, Zrivada Analogs
Zrivada Brand Names Mixture
- No information avaliable
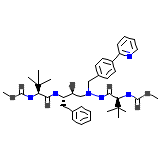
Zrivada Chemical_Formula
C38H52N6O7
Zrivada RX_link
http://www.rxlist.com/cgi/generic3/reyataz.htm
Zrivada fda sheet
Zrivada msds (material safety sheet)
Zrivada Synthesis Reference
No information avaliable
Zrivada Molecular Weight
704.856 g/mol
Zrivada Melting Point
No information avaliable
Zrivada H2O Solubility
Free base slightly soluble (4-5 mg/mL)
Zrivada State
Solid
Zrivada LogP
4.25
Zrivada Dosage Forms
Capsule (100 mg, 150 mg, or 200 mg)
Zrivada Indication
For use, in combination with other antiretroviral agents, in the treatment of HIV-1 infection.
Zrivada Pharmacology
Atazanavir (ATV) is an azapeptide HIV-1 protease inhibitor (PI). It is a protease inhibitor with activity against Human Immunodeficiency Virus Type 1 (HIV-1). Protease inhibitors block the part of HIV called protease. HIV-1 protease is an enzyme required for the proteolytic cleavage of the viral polyprotein precursors into the individual functional proteins found in infectious HIV-1. Atazanavir binds to the protease active site and inhibits the activity of the enzyme. This inhibition prevents cleavage of the viral polyproteins resulting in the formation of immature non-infectious viral particles. Protease inhibitors are almost always used in combination with at least two other anti-HIV drugs.
Zrivada Absorption
Atazanavir is rapidly absorbed with a Tmax of approximately 2.5 hours. Administration of atazanavir with food enhances bioavailability and reduces pharmacokinetic variability
Zrivada side effects and Toxicity
No information avaliable
Zrivada Patient Information
Zrivada Organisms Affected
Human immunodeficiency virus