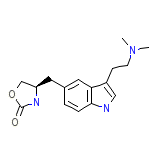
Zomig




Zomig Brand names, Zomig Analogs
Zomig Brand Names Mixture
- No information avaliable
Zomig Chemical_Formula
C16H21N3O2
Zomig RX_link
http://www.rxlist.com/cgi/generic2/zolmit.htm
Zomig fda sheet
Zomig msds (material safety sheet)
Zomig Synthesis Reference
No information avaliable
Zomig Molecular Weight
287.357 g/mol
Zomig Melting Point
No information avaliable
Zomig H2O Solubility
No information avaliable
Zomig State
Solid
Zomig LogP
1.363
Zomig Dosage Forms
Spray; Tablet
Zomig Indication
For the acute treatment of adult migraine (with or without auras)
Zomig Pharmacology
Zolmitriptan is a selective 5-hydroxytryptamine receptor subtype agonist indicated for the acute treatment of migraine attacks with or without aura in adults. Zolmitriptan is not intended for the prophylactic therapy of migraine or for use in the management of hemiplegic or basilar migraine. Zolmitriptan is an agonist for a vascular 5-hydroxytryptamine receptor subtype (probably a member of the 5-HT1D family) having only a weak affinity for 5-HT1A, 5-HT5A, and 5-HT7 receptors and no significant affinity or pharmacological activity at 5-HT2, 5-HT3 or 5-HT4 receptor subtypes or at alpha1-, alpha2-, or beta-adrenergic, dopamine1,; dopamine2; muscarinic, or benzodiazepine receptors. This action in humans correlates with the relief of migraine headache. In addition to causing vasoconstriction, experimental data from animal studies show that Zolmitriptan also activates 5-HT1 receptors on peripheral terminals of the trigeminal nerve innervating cranial blood vessels, which may also contribute to the antimigrainous effect of Zolmitriptan in humans.
Zomig Absorption
40%
Zomig side effects and Toxicity
No information avaliable
Zomig Patient Information
No information avaliable
Zomig Organisms Affected
Humans and other mammals