Vermisol 150




Vermisol 150 Brand names, Vermisol 150 Analogs
Vermisol 150 Brand Names Mixture
- No information avaliable
Vermisol 150 Chemical_Formula
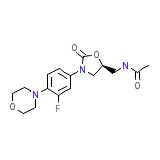
C16H20FN3O4
Vermisol 150 RX_link
http://www.rxlist.com/cgi/generic3/linezolid.htm
Vermisol 150 fda sheet
Vermisol 150 msds (material safety sheet)
Vermisol 150 Synthesis Reference
No information avaliable
Vermisol 150 Molecular Weight
337.346 g/mol
Vermisol 150 Melting Point
No information avaliable
Vermisol 150 H2O Solubility
3 mg/mL
Vermisol 150 State
Solid
Vermisol 150 LogP
0.232
Vermisol 150 Dosage Forms
Solution; Tablets
Vermisol 150 Indication
For the treatment of bacterial infections caused by susceptible strains of vancomycin resistant Enterococcus faecium, Staphylococcal aureus (methicillin resistant and susceptible strains), Streptococcus pneumoniae, Streptococcus pyogenes, Streptococcus agalactiae.
Vermisol 150 Pharmacology
Linezolid is a synthetic antibacterial agent of a new class of antibiotics, the oxazolidinones, which has clinical utility in the treatment of infections caused by aerobic Gram-positive bacteria. The in vitro spectrum of activity of linezolid also includes certain Gram-negative bacteria and anaerobic bacteria. Susceptible organisms include methicillin- and vancomycin-resistant staphylococci, vancomycin-resistant enterococci, penicillin-resistant pneumococci and anaerobes. Oxazolidinones inhibit protein synthesis by binding at the P site at the ribosomal 50S subunit. Resistance to other protein synthesis inhibitors does not affect oxazolidinone activity, however rare development of oxazolidinone resistance cases, associated with 23S rRNA alterations during treatment have been reported. Linezolid inhibits bacterial protein synthesis through a mechanism of action different from that of other antibacterial agents; therefore, cross-resistance between linezolid and other classes of antibiotics is unlikely.
Vermisol 150 Absorption
Linezolid is rapidly and extensively absorbed after oral dosing. Maximum plasma concentrations are reached approximately 1 to 2 hours after dosing, and the absolute bioavailability is approximately 100%.
Vermisol 150 side effects and Toxicity
Clinical signs of acute toxicity lead to decreased activity, ataxia, vomiting and tremors.
Vermisol 150 Patient Information
Vermisol 150 Organisms Affected
Gram negative and gram positive bacteria