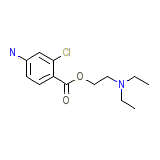
Ultracaine D-S




Ultracaine D-S Brand names, Ultracaine D-S Analogs
- Astracaine 4%
- Astracaine 4% Forte
- Carbocaine
- Carbocaine with Neo-Cobefrin
- Chirocaine
- Chlor-Procaine
- Chloroprocain
- Chloroprocaine Hcl
- Chloroprocainum [Inn-Latin]
- Chlorprocaine
- Chlorprocainum
- Citanest Forte
- Citanest Plain
- Cloroprocaina [Inn-Spanish]
- Dalcaine
- Dilocaine
- Duranest
- Duranest-Mpf
- Halestyn
- Isocaine
- Isocaine 2%
- Isocaine 3%
- L-Caine
- Lidoject-1
- Lidoject-2
- Marcaine
- Marcaine Spinal
- Nesacaine
- Nesacaine-Ce
- Nesacaine-Mpf
- Novocain
- Octocaine
- Octocaine-100
- Octocaine-50
- Piocaine
- Polocaine
- Polocaine-Mpf
- Pontocaine
- Sensorcaine
- Sensorcaine Forte
- Sensorcaine-Mpf
- Sensorcaine-Mpf Spinal
- Septocaine
- Ultracaine D-S
- Ultracaine D-S Forte
- Xylocaine
- Xylocaine 5% Spinal
- Xylocaine Test Dose
- Xylocaine-Mpf
- Xylocaine-Mpf with Glucose
Ultracaine D-S Brand Names Mixture
- No information avaliable
Ultracaine D-S Chemical_Formula
C13H19ClN2O2
Ultracaine D-S RX_link
http://www.rxlist.com/cgi/generic3/chloroprocaine.htm
Ultracaine D-S fda sheet
Ultracaine D-S msds (material safety sheet)
Ultracaine D-S Synthesis Reference
No information avaliable
Ultracaine D-S Molecular Weight
270.755 g/mol
Ultracaine D-S Melting Point
No information avaliable
Ultracaine D-S H2O Solubility
665 mg/L
Ultracaine D-S State
Solid
Ultracaine D-S LogP
2.464
Ultracaine D-S Dosage Forms
Liquid
Ultracaine D-S Indication
For the production of local anesthesia by infiltration and peripheral nerve block. They are not to be used for lumbar or caudal epidural anesthesia.
Ultracaine D-S Pharmacology
Chloroprocaine is an anesthetic agent indicated for production of local or regional anesthesia, particularly for oral surgery. Chloroprocaine (like cocaine) has the advantage of constricting blood vessels which reduces bleeding, unlike other local anesthetics like lidocaine. Chloroprocaine is an ester anesthetic.
Ultracaine D-S Absorption
No information avaliable
Ultracaine D-S side effects and Toxicity
No information avaliable
Ultracaine D-S Patient Information
When appropriate, patients should be informed in advance that they may experience temporary loss of sensation and motor activity, usually in the lower half of the body, following proper administration of epidural anesthesia.
Ultracaine D-S Organisms Affected
Humans and other mammals