Masteron




Masteron Brand names, Masteron Analogs
Masteron Brand Names Mixture
- No information avaliable
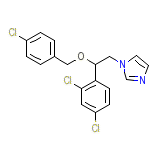
Masteron Chemical_Formula
C18H15Cl3N2O
Masteron RX_link
http://www.rxlist.com/cgi/generic3/econazole.htm
Masteron fda sheet
Masteron msds (material safety sheet)
Masteron Synthesis Reference
No information avaliable
Masteron Molecular Weight
381.683 g/mol
Masteron Melting Point
No information avaliable
Masteron H2O Solubility
No information avaliable
Masteron State
No information avaliable
Masteron LogP
5.401
Masteron Dosage Forms
Topical cream (1%); Suppository
Masteron Indication
For topical application in the treatment of tinea pedis, tinea cruris, and tinea corporis caused by Trichophyton rubrum, Trichophyton mentagrophytes, Trichophyton tonsurans, Microsporum canis, Microsporum audouini, Microsporum gypseum, and Epidermophyton floccosum, in the treatment of cutaneous candidiasis, and in the treatment of tinea versicolor.
Masteron Pharmacology
Econazole is an antifungal medication related to fluconazole (Diflucan), ketoconazole (Nizoral), itraconazole (Sporanox), and clotrimazole (Lotrimin, Mycelex). Econazole prevents fungal organisms from producing vital substances required for growth and function. This medication is effective only for infections caused by fungal organisms. It will not work for bacterial or viral infections.
Masteron Absorption
After topical application to the skin of normal subjects, systemic absorption of econazole nitrate is extremely low. Although most of the applied drug remains on the skin surface, drug concentrations were found in the stratum corneum which, by far, exceeded the minimum inhibitory concentration for dermatophytes.
Masteron side effects and Toxicity
Overdosage of econazole in humans has not been reported to date. In mice, rats guinea pigs and dogs, the oral LD 50 values were found to be 462, 668, 272, and > 160 mg/kg, respectively.
Masteron Patient Information
No information avaliable
Masteron Organisms Affected
Yeast and other fungi