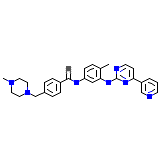
Imatinib




Imatinib Brand names, Imatinib Analogs
Imatinib Brand Names Mixture
- No information avaliable
Imatinib Chemical_Formula
C29H31N7O
Imatinib RX_link
http://www.rxlist.com/cgi/generic3/gleevec.htm
Imatinib fda sheet
Imatinib msds (material safety sheet)
Imatinib Synthesis Reference
No information avaliable
Imatinib Molecular Weight
493.603 g/mol
Imatinib Melting Point
226 oC (mesylate salt)
Imatinib H2O Solubility
Very soluble in water at pH < 5.5 (mesylate salt)
Imatinib State
Solid
Imatinib LogP
3.218
Imatinib Dosage Forms
Capsule; Tablet
Imatinib Indication
For the treatment of newly diagnosed adult patients with Philadelphia chromosome positive chronic myeloid leukemia (CML). Also indicated for the treatment of pediatric patients with Ph+ chronic phase CML whose disease has recurred after stem cell transplant or who are resistant to interferon-alpha therapy. Also indicated with unresectable and/or metastatic malignant gastrointestinal stromal tumors (GIST).
Imatinib Pharmacology
Imatinib is an antineoplastic agent used to treat chronic myelogenous leukemia. Imatinib is a 2-phenylaminopyrimidine derivative that functions as a specific inhibitor of a number of tyrosine kinase enzymes. In chronic myelogenous leukemia, the Philadelphia chromosome leads to a fusion protein of Abl with Bcr (breakpoint cluster region), termed Bcr-Abl. As this is now a continuously active tyrosine kinase, Imatinib is used to decrease Bcr-Abl activity.
Imatinib Absorption
Imatinib is well absorbed with mean absolute bioavailability is 98% with maximum levels achieved within 2-4 hours of dosing
Imatinib side effects and Toxicity
Side effects include nausea, vomiting, diarrhea, loss of appetite, dry skin, hair loss, swelling (especially in the legs or around the eyes) and muscle cramps
Imatinib Patient Information
Imatinib Organisms Affected
Humans and other mammals