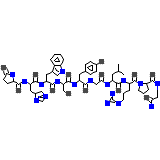
Gonadorelin




Gonadorelin Brand names, Gonadorelin Analogs
Gonadorelin Brand Names Mixture
- No information avaliable
Gonadorelin Chemical_Formula
C55H75N17O13
Gonadorelin RX_link
http://www.rxlist.com/cgi/generic2/gonadorelin.htm
Gonadorelin fda sheet
Gonadorelin msds (material safety sheet)
Gonadorelin Synthesis Reference
No information avaliable
Gonadorelin Molecular Weight
1182.29 g/mol
Gonadorelin Melting Point
No information avaliable
Gonadorelin H2O Solubility
1 mg/mL
Gonadorelin State
Liquid
Gonadorelin LogP
-4.634
Gonadorelin Dosage Forms
Liquid; Powder for solution; Solution for infusion pump
Gonadorelin Indication
For evaluating the functional capacity and response of the gonadotropes of the anterior pituitary also for evaluating residual gonadotropic function of the pituitary following removal of a pituitary tumor by surgery and/or irradiation.
Gonadorelin Pharmacology
Gonadorelin is responsible for the release of follicle stimulating hormone and leutinizing hormone from the anterior pitutitary. In the pituitary GnRH stimulates synthesis and release of FSH and LH, a process that is controlled by the frequency and amplitude of GnRH pulses, as well as the feedback of androgens and estrogens. The pulsatility of GnRH secretion has been seen in all vertebrates, and it is necessary to ensure a correct reproductive function. Thus a single hormone, GnRH, controls a complex process of follicular growth, ovulation, and corpus luteum maintenance in the female, and spermatogenesis in the male. Its short half life requires infusion pumps for its clinical use
Gonadorelin Absorption
Rapidly absorbed when injected
Gonadorelin side effects and Toxicity
LD50>3000 mg/kg (rat, oral)
Gonadorelin Patient Information
Gonadorelin Organisms Affected
Humans and other mammals