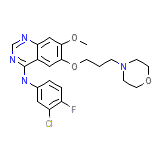
Gefitinib




Gefitinib Brand names, Gefitinib Analogs
Gefitinib Brand Names Mixture
- No information avaliable
Gefitinib Chemical_Formula
C22H24ClFN4O3
Gefitinib RX_link
http://www.rxlist.com/cgi/generic3/iressa.htm
Gefitinib fda sheet
Gefitinib msds (material safety sheet)
Gefitinib Synthesis Reference
No information avaliable
Gefitinib Molecular Weight
446.902 g/mol
Gefitinib Melting Point
No information avaliable
Gefitinib H2O Solubility
Sparingly soluble (<pH4)
Gefitinib State
Solid
Gefitinib LogP
3.46
Gefitinib Dosage Forms
Tablet
Gefitinib Indication
For the continued treatment of patients with locally advanced or metastatic non-small cell lung cancer after failure of either platinum-based or docetaxel chemotherapies.
Gefitinib Pharmacology
Gefitinib inhibits the intracellular phosphorylation of numerous tyrosine kinases associated with transmembrane cell surface receptors, including the tyrosine kinases associated with the epidermal growth factor receptor (EGFR-TK). EGFR is expressed on the cell surface of many normal cells and cancer cells.
Gefitinib Absorption
Absorbed slowly after oral administration with mean bioavailability of 60%.
Gefitinib side effects and Toxicity
No information avaliable
Gefitinib Patient Information
Gefitinib Organisms Affected
Humans and other mammals