Finastid




Finastid Brand names, Finastid Analogs
Finastid Brand Names Mixture
- No information avaliable
Finastid Chemical_Formula
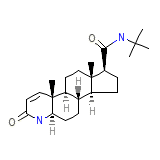
C23H36N2O2
Finastid RX_link
http://www.rxlist.com/cgi/generic3/propecia.htm
Finastid fda sheet
Finastid msds (material safety sheet)
Finastid Synthesis Reference
No information avaliable
Finastid Molecular Weight
372.544 g/mol
Finastid Melting Point
252-254 oC
Finastid H2O Solubility
11.7 mg/L
Finastid State
Solid
Finastid LogP
4.277
Finastid Dosage Forms
Tablet
Finastid Indication
For the treatment of symptomatic benign prostatic hyperplasia (BPH) in men with an enlarged prostate to: Improve symptoms, reduce the risk of acute urinary retention, reduce the risk of the need for surgery including transurethral resection of the prostat.
Finastid Pharmacology
Finasteride is indicated for the treatment of male pattern hair loss (androgenetic alopecia) in men only. Finasteride is a competitive and specific inhibitor of Type II 5a-reductase, an intracellular enzyme that converts the androgen testosterone into DHT. Two distinct isozymes are found in mice, rats, monkeys, and humans: Type I and II. Each of these isozymes is differentially expressed in tissues and developmental stages. In humans, Type I 5a-reductase is predominant in the sebaceous glands of most regions of skin, including scalp, and liver. Type I 5a-reductase is responsible for approximately one-third of circulating DHT. The Type II 5a-reductase isozyme is primarily found in prostate, seminal vesicles, epididymides, and hair follicles as well as liver, and is responsible for two-thirds of circulating DHT.
Finastid Absorption
No information avaliable
Finastid side effects and Toxicity
No information avaliable
Finastid Patient Information
No information avaliable
Finastid Organisms Affected
Humans and other mammals