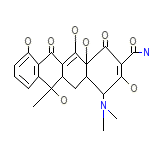
Cyclopar




Cyclopar Brand names, Cyclopar Analogs
- Abramycin
- Abricycline
- Achromycin
- Achromycin V
- Achromycin, Naphthacene Derivative
- Actisite
- Agromicina
- Ambramicina
- Ambramycin
- Amycin
- Anhydrotetracycline
- Aureomycin
- Bio-Tetra
- Biocycline
- Bristaciclin
- Bristaciclin α
- Bristaciclina
- Bristacycline
- Cefracycline
- Ciclibion
- Copharlan
- Criseociclina
- Cyclomycin
- Cyclopar
- Cytome
- Democracin
- Deschlorobiomycin
- Dumocyclin
- Enterocycline
- Hostacyclin
- Lexacycline
- Limecycline
- Liquamycin
- Medocycline
- Mericycline
- Micycline
- Neocycline
- Oletetrin
- Omegamycin
- Orlycycline
- Oxytetracycline HCl
- Panmycin
- Polycycline
- Polyotic
- Purocyclina
- Resteclin
- Retet
- Robitet
- Roviciclina
- SK-Tetracycline
- Solvocin
- Sumycin
- Sumycin Syrup
- TAC
- TC
- Terramycin
- Tetra-CO
- Tetrabon
- Tetrachel
- Tetracycl
- Tetracycline HCl
- Tetracycline II
- Tetracyn
- Tetradecin
- Tetrafil
- Tetramed
- Tetraverine
- Tetrex
- Topicycline
- Tsiklomistsin
- Tsiklomitsin
- Veracin
- Vetacyclinum
Cyclopar Brand Names Mixture
- Delta-Albaplex tablets (Novobiocin sodium + Prednisolone + Tetracycline hydrochloride)
- Neo Chlor PWR (Neomycin sulfate + Tetracycline hydrochloride)
- Neo-Chlor (Neomycin sulfate + Tetracycline hydrochloride)
- Neotetramed PWR (Neomycin sulfate + Tetracycline hydrochloride)
- P Onytricin P (Calcium D-pantothenate + Menadione sodium bisulfite + Nicotinamide + Tetracycline hydrochloride + Vitamin A + Vitamin B12 + Vitamin B2 + Vitamin B6 + Vitamin D3 + Vitamin E)
Cyclopar Chemical_Formula
C22H24N2O8
Cyclopar RX_link
http://www.rxlist.com/cgi/generic2/tetcycl2.htm
Cyclopar fda sheet
Cyclopar msds (material safety sheet)
Cyclopar Synthesis Reference
J. H. Boothe et al., J. Am. Chem. Soc. 75, 4621 (1953)
Cyclopar Molecular Weight
444.435 g/mol
Cyclopar Melting Point
165oC
Cyclopar H2O Solubility
231 mg/L at 25 oC (SRC PhysProp experimental -- YALKOWSKY,SH & DANNENFELSER,RM (1992))
Cyclopar State
Solid
Cyclopar LogP
-0.304
Cyclopar Dosage Forms
Tablets; Oral suspension; Capsules; Topical solution; Ophthalmic suspension; IV injection; IM injection; Ointment
Cyclopar Indication
Used to treat bacterial infections such as Rocky Mountain spotted fever, typhus fever, and tick fevers; upper respiratory infections; pneumonia; gonorrhea; amoebic infections; and urinary tract infections. It is also used to help treat severe acne and to treat trachoma (a chronic eye infection) and conjunctivitis (pinkeye). Tetracycline is often an alternative drug for people who are allergic to penicillin.
Cyclopar Pharmacology
Tetracycline is a short-acting antibiotic semisynthetically produced from chlortetracycline, which is derived from Streptomyces aureofaciens. Tetracycline inhibits cell growth by inhibiting translation. It binds to the 30S ribosomal subunit and prevents the amino-acyl tRNA from binding to the A site of the ribosome. This binding is reversible in nature.
Cyclopar Absorption
Bioavailability is less than 40% when administered via intramuscular injection, 100% intravenously, and 60-80% orally (fasting adults). Food and/or milk reduce GI absorption of oral preparations of tetracycline by 50% or more.
Cyclopar side effects and Toxicity
LD50=808mg/kg (orally in mice)
Cyclopar Patient Information
Cyclopar Organisms Affected
Enteric bacteria and other eubacteria