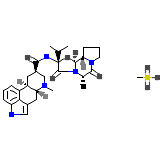
Co-Dergocrine Mesylate




Co-Dergocrine Mesylate Brand names, Co-Dergocrine Mesylate Analogs
- Alkergot
- Circanol
- Co-Dergocrine Mesylate
- Deapril-ST
- Dihydroergotoxin Mesilat
- Dihydroergotoxin Mesylate
- Dihydroergotoxin Methanesulfonate
- Dihydroergotoxine Mesilate
- Dihydroergotoxine Mesylate
- Dihydroergotoxine Methanesulfonate
- Dihydroergotoxine Methanesulphonate
- Ergoloid Mesylates [Usan]
- Gerimal
- Hydergin
- Hydergine
- Hydergine LC
- Hydrogenated Ergot Alkaloids
- Ischelium
- Redergin
- Trigot
Co-Dergocrine Mesylate Brand Names Mixture
- No information avaliable
Co-Dergocrine Mesylate Chemical_Formula
C33H45N5O5
Co-Dergocrine Mesylate RX_link
No information avaliable
Co-Dergocrine Mesylate fda sheet
Co-Dergocrine Mesylate msds (material safety sheet)
Co-Dergocrine Mesylate Synthesis Reference
No information avaliable
Co-Dergocrine Mesylate Molecular Weight
591.741 g/mol
Co-Dergocrine Mesylate Melting Point
No information avaliable
Co-Dergocrine Mesylate H2O Solubility
No information avaliable
Co-Dergocrine Mesylate State
Solid
Co-Dergocrine Mesylate LogP
2.615
Co-Dergocrine Mesylate Dosage Forms
Capsule; Liquid; Powder; Powder for solution; Solution; Syrup; Tablet
Co-Dergocrine Mesylate Indication
For use as an adjunct therapy for patients with dementia
Co-Dergocrine Mesylate Pharmacology
Ergoloid Mesylate may increase cerebral metabolism and blood flow. The role of this medication in the therapy of dementia is controversial. A recent controlled study in patients with Alzheimer's disease found that there was no advantage to the use of ergoloid mesylates compared to placebo, suggesting that ergoloid mesylates may lower scores on some cognitive and behavioral rating scales. Further study is needed to determine the risk-benefit profile of ergoloid mesylates in the treatment of dementia.
Co-Dergocrine Mesylate Absorption
Rapidly but incompletely (approximately 25%) absorbed from the gastrointestinal tract. Approximately 50% of the absorbed dose is eliminated by first-pass metabolism.
Co-Dergocrine Mesylate side effects and Toxicity
Symptoms of overdose include dyspnea, hypotension or hypertension, rapid weak pulse, delirium, nausea, vomiting, and bradycardia.
Co-Dergocrine Mesylate Patient Information
No information avaliable
Co-Dergocrine Mesylate Organisms Affected
Humans and other mammals