CholestaGel




CholestaGel Brand names, CholestaGel Analogs
CholestaGel Brand Names Mixture
- No information avaliable
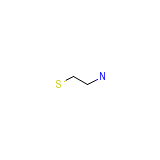
CholestaGel Chemical_Formula
C2H7NS
CholestaGel RX_link
No information avaliable
CholestaGel fda sheet
CholestaGel msds (material safety sheet)
CholestaGel Synthesis Reference
No information avaliable
CholestaGel Molecular Weight
77.1497 g/mol
CholestaGel Melting Point
98 oC
CholestaGel H2O Solubility
No information avaliable
CholestaGel State
Solid
CholestaGel LogP
-0.196
CholestaGel Dosage Forms
Capsule (50 mg, 150 mg)
CholestaGel Indication
Given intravenously or orally to treat radiation sickness. The bitartrate has been used for the oral treatment of nephropathic cystinosis.
CholestaGel Pharmacology
People born without the ability to metabolize the amino acid cystine suffer from cystinosis, a rare inherited disorder characterized by the deposition and accumulation of cystine crystals throughout the body. These crystals cause considerable damage, particularly in the kidney. Kidney failure can occur by the age of 10 in untreated patients. Cysteamine prevents the accumulation of cystine crystals and is prescribed to prevent further kidney damage. Cysteamine helps to convert cystine into less harmful chemical forms that can be removed from cells.
CholestaGel Absorption
No information avaliable
CholestaGel side effects and Toxicity
Symptoms of overdose may include convulsions (seizures), increased thirst and unusual tiredness or weakness.
CholestaGel Patient Information
No information avaliable
CholestaGel Organisms Affected
Humans and other mammals