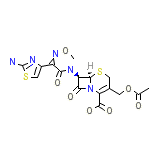
Cefotaxime sodium




Cefotaxime sodium Brand names, Cefotaxime sodium Analogs
Cefotaxime sodium Brand Names Mixture
- No information avaliable
Cefotaxime sodium Chemical_Formula
C16H17N5O7S2
Cefotaxime sodium RX_link
http://www.rxlist.com/cgi/generic/cefotax.htm
Cefotaxime sodium fda sheet
Cefotaxime sodium msds (material safety sheet)
Cefotaxime sodium Synthesis Reference
No information avaliable
Cefotaxime sodium Molecular Weight
455.468 g/mol
Cefotaxime sodium Melting Point
No information avaliable
Cefotaxime sodium H2O Solubility
Soluble
Cefotaxime sodium State
Solid
Cefotaxime sodium LogP
-0.685
Cefotaxime sodium Dosage Forms
Injection and infusion
Cefotaxime sodium Indication
Used to treat gonorrhoea, meningitis, and severe infections including infections of the kidney (pyelonephritis) and urinary system. Also used before an operation to prevent infection after surgery.
Cefotaxime sodium Pharmacology
Cefotaxime is a third generation intravenous cephalosporin antibiotic. It has broad spectrum activity against Gram positive and Gram negative bacteria. It does not have activity against Pseudomonas aeruginosa. Cefotaxime works by inhibiting bacterial cell wall biosynthesis. A positive feature of cefotaxime is that it display a resistance to penicillinases and is useful to treat infections that are resistant to penicillin derivatives.
Cefotaxime sodium Absorption
Rapidly absorbed following intramuscular injection.
Cefotaxime sodium side effects and Toxicity
Adverse effects following overdosage include nausea, vomiting, epigastric distress, diarrhea, and convulsions. Oral rat LD50 is over 20,000 mg/kg while intravenous rat LD50 is over 7,000 mg/kg.
Cefotaxime sodium Patient Information
No information avaliable
Cefotaxime sodium Organisms Affected
Enteric bacteria and other eubacteria