Avelox




Avelox Brand names, Avelox Analogs
Avelox Brand Names Mixture
- No information avaliable
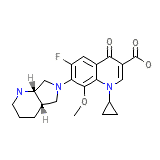
Avelox Chemical_Formula
C21H24FN3O4
Avelox RX_link
http://www.rxlist.com/cgi/generic3/vigamox.htm
Avelox fda sheet
Avelox msds (material safety sheet)
Avelox Synthesis Reference
No information avaliable
Avelox Molecular Weight
401.431 g/mol
Avelox Melting Point
238-242 oC
Avelox H2O Solubility
No information avaliable
Avelox State
Solid
Avelox LogP
2.033
Avelox Dosage Forms
Solution (250 mL bags containing 400 mg of drug); Tablets (400 mg)
Avelox Indication
For the treatment of sinus and lung infections such as sinusitis, pneumonia, and secondary infections in chronic bronchitis. Also for the treatment of bacterial conjunctivitis (pinkeye).
Avelox Pharmacology
Moxifloxacin is a quinolone/fluoroquinolone antibiotic. Moxifloxacin can be used to treat infections caused by the following bacteria: Aerobic Gram-positive microorganisms: Corynebacterium species, Micrococcus luteus, Staphylococcus aureus, Staphylococcus epidermidis, Staphylococcus haemolyticus, Staphylococcus hominis, Staphylococcus warneri, Streptococcus pneumoniae, and Streptococcus viridans group. Aerobic Gram-negative microorganisms: Acinetobacter lwoffii, Haemophilus influenzae, and Haemophilus parainfluenzae. Other microorganisms: Chlamydia trachomatis.
Moxifloxacin is bactericidal and its mode of action depends on blocking of bacterial DNA replication by binding itself to an enzyme called DNA gyrase, which allows the untwisting required to replicate one DNA double helix into two. Notably the drug has 100 times higher affinity for bacterial DNA gyrase than for mammalian. Moxifloxacin is a broad-spectrum antibiotic that is active against both Gram-positive and Gram-negative bacteria.
Moxifloxacin is bactericidal and its mode of action depends on blocking of bacterial DNA replication by binding itself to an enzyme called DNA gyrase, which allows the untwisting required to replicate one DNA double helix into two. Notably the drug has 100 times higher affinity for bacterial DNA gyrase than for mammalian. Moxifloxacin is a broad-spectrum antibiotic that is active against both Gram-positive and Gram-negative bacteria.
Avelox Absorption
Well absorbed from the gastrointestinal tract. Absolute oral bioavailability is approximately 90%. Food has little effect on absorption.
Avelox side effects and Toxicity
Symptoms of overdose include CNS and gastrointestinal effects such as decreased activity, somnolence, tremor, convulsions, vomiting, and diarrhea. The minimal lethal intravenous dose in mice and rats is 100 mg/kg.
Avelox Patient Information
Avoid contaminating the applicator tip with material from the eye, fingers or other source. Systemically administered quinolones including moxifloxacin have been associated with hypersensitivity reactions, even following a single dose. Discontinue use immediately and contact your physician at the first sign of a rash or allergic reaction.
Avelox Organisms Affected
Enteric bacteria and other eubacteria