Zydis




Categoria
Zydis Marchi, Zydis Analoghi
Zydis Marchi miscela
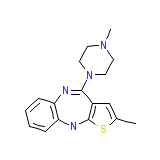
Zydis Formula chimica
C17H20N4S
Zydis RX link
http://www.rxlist.com/cgi/generic3/symbyax.htm
Zydis FDA foglio
Zydis DMS (foglio di materiale di sicurezza)
Zydis Sintesi di riferimento
Chakrabarti JK et al. US Pat. 5,229,382 (1991)
Zydis Peso molecolare
312.434 g/mol
Zydis Temperatura di fusione
195oC
Zydis H2O Solubilita
Nessuna informazione disponibile
Zydis Stato
Solid
Zydis LogP
2.199
Zydis Forme di dosaggio
Tablet (orale)
Zydis Indicazione
Per il trattamento della schizofrenia e psicosi maniaco-depressiva (disturbo bipolare).
Zydis Farmacologia
Olanzapina, un farmaco antipsicotico atipico, è usata per trattare sia i sintomi positivi e negativi della schizofrenia, mania acuta con disturbo bipolare, agitazione, sintomi psicotici e nella demenza. Futuro gli usi possono includere il trattamento del disturbo ossessivo-compulsivo e gravi disturbi comportamentali nell'autismo. Strutturalmente e farmacologicamente simile alla clozapina, olanzapina si lega per alfa (1), la dopamina, , H1 dell'istamina, muscarinici, e la serotonina di tipo 2 (5-HT2) recettori.
Zydis Assorbimento
Ben assorbito, con circa il 40% della dose è metabolizzato prima di raggiungere la circolazione sistemica.
Zydis Tossicita
Nessuna informazione disponibile
Zydis Informazioni paziente
PATIENT INFORMATION
Physicians are advised to discuss the following issues with patients for whom they prescribe
olanzapine:
Orthostatic Hypotension: Patients should be advised of the risk of orthostatic hypotension,
especially during the period of initial dose titration and in association with the use of
concomitant drugs that may potentiate the orthostatic effect of olanzapine, e.g., diazepam
or alcohol.
Interference with Cognitive and Motor Performance: Because olanzapine has the potential to
impair judgment, thinking, or motor skills, patients should be cautioned about operating
hazardous machinery, including automobiles, until they are reasonably certain that olanzapine
therapy does not affect them adversely.
Pregnancy: Patients should be advised to notify their physician if they become pregnant or
intend to become pregnant during therapy with olanzapine.
Nursing: Patients should be advised not to breast-feed an infant if they are taking olanzapine.
Concomitant Medication: Patients should be advised to inform their physicians if they are taking,
or plan to take, any prescription or over-the-counter drugs, since there is a potential for
interactions.
Alcohol: Patients should be advised to avoid alcohol while taking olanzapine.
Heat Exposure and Dehydration: Patients should be advised regarding appropriate care in avoiding
overheating and dehydration.
Phenylketonurics: ZYPREXA ZYDIS (olanzapine orally disintegrating tablets) contains phenylalanine
(0.34, 0.45, 0.67, or 0.90 mg per 5, 10, 15, or 20mg tablet, respectively).
Laboratory Tests
Periodic assessment of transaminases is recommended in patients with significant hepatic disease.
Physicians are advised to discuss the following issues with patients for whom they prescribe
olanzapine:
Orthostatic Hypotension: Patients should be advised of the risk of orthostatic hypotension,
especially during the period of initial dose titration and in association with the use of
concomitant drugs that may potentiate the orthostatic effect of olanzapine, e.g., diazepam
or alcohol.
Interference with Cognitive and Motor Performance: Because olanzapine has the potential to
impair judgment, thinking, or motor skills, patients should be cautioned about operating
hazardous machinery, including automobiles, until they are reasonably certain that olanzapine
therapy does not affect them adversely.
Pregnancy: Patients should be advised to notify their physician if they become pregnant or
intend to become pregnant during therapy with olanzapine.
Nursing: Patients should be advised not to breast-feed an infant if they are taking olanzapine.
Concomitant Medication: Patients should be advised to inform their physicians if they are taking,
or plan to take, any prescription or over-the-counter drugs, since there is a potential for
interactions.
Alcohol: Patients should be advised to avoid alcohol while taking olanzapine.
Heat Exposure and Dehydration: Patients should be advised regarding appropriate care in avoiding
overheating and dehydration.
Phenylketonurics: ZYPREXA ZYDIS (olanzapine orally disintegrating tablets) contains phenylalanine
(0.34, 0.45, 0.67, or 0.90 mg per 5, 10, 15, or 20mg tablet, respectively).
Laboratory Tests
Periodic assessment of transaminases is recommended in patients with significant hepatic disease.
Zydis Atto interessato organismi
Gli esseri umani e altri mammiferi