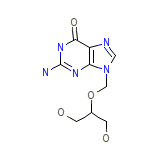
GCV




Categoria
GCV Marchi, GCV Analoghi
GCV Marchi miscela
GCV Formula chimica
C9H13N5O4
GCV RX link
http://www.rxlist.com/cgi/generic2/vitrasert.htm
GCV FDA foglio
GCV DMS (foglio di materiale di sicurezza)
GCV Sintesi di riferimento
Alhede, Boerge, et al;. J.Org.Chem;. 56, 6; 2139-2143 (1991)
GCV Peso molecolare
255.231 g/mol
GCV Temperatura di fusione
250 oC
GCV H2O Solubilita
4,3 mg / ml
GCV Stato
Solid
GCV LogP
-1.438
GCV Forme di dosaggio
Occular inserire; capsula orale, iniezione
GCV Indicazione
Per l'induzione e di mantenimento nel trattamento del citomegalovirus (CMV) retinite nei pazienti immunocompromessi, inclusi i pazienti con sindrome da immunodeficienza acquisita (AIDS). Utilizzato anche nel trattamento di gravi citomegalovirus (CMV) malattia, compresa la polmonite CMV, malattia da CMV gastrointestinali, e diffuse infezioni da CMV nei pazienti immunocompromessi.
GCV Farmacologia
Ganciclovir è un analogo nucleosidico sintetico della 2'-deossiguanosina che inibisce la replicazione del virus herpes sia in vitro che in vivo. Virus umani sono sensibili citomegalovirus (CMV), herpes virus di simplex di -1 e -2 (HSV-1, HSV-2), virus Epstein-Barr (EBV) e del virus della varicella zoster (VZV), tuttavia gli studi clinici sono stati limitati alla valutazione di efficacia nei pazienti con infezione da CMV. Ganciclovir è un profarmaco che è strutturalmente simile alla aciclovir. Inibisce la replicazione del virus dal suo encorporation nel DNA virale. Questo encorporation inibisce dATP e conduce ad un difetto del DNA, arrestare o ritardare il meccanismo virale necessario per diffondere il virus ad altre cellule.
GCV Assorbimento
Scarsamente assorbito per via sistemica dopo somministrazione orale. La biodisponibilità in condizioni di digiuno è di circa il 5%, e quando somministrato con il cibo, da 6 a 9% (circa il 30% con un pasto grasso).
GCV Tossicita
Orale, mouse LD50:> 2g/kg. Per via endovenosa, il cane LD50: 150mg/kg>. Sintomi di sovradosaggio includono pancitopenia irreversibile, peggioramento dei sintomi gastrointestinali e insufficienza renale acuta. Agente di cancro sospetto.
GCV Informazioni paziente
All patients should be informed that the major toxicities of ganciclovir are granulocytopenia
(neutropenia), anemia and thrombocytopenia and that dose modifications may be required, including
discontinuation. The importance of close monitoring of blood counts while on therapy should be emphasized.
Patients should be informed that ganciclovir has been associated with elevations in serum creatinine.
Patients should be instructed to take CYTOVENE capsules with food to maximize bioavailability.
Patients should be advised that ganciclovir has caused decreased sperm production in animals and may cause
infertility in humans. Women of childbearing potential should be advised that ganciclovir causes birth
defects in animals and should not be used during pregnancy. Women of childbearing potential should be
advised to use effective contraception during treatment with CYTOVENE-IV or CYTOVENE. Similarly, men
should be advised to practice barrier contraception during and for at least 90 days following treatment
with CYTOVENE-IV or CYTOVENE.
Patients should be advised that ganciclovir causes tumors in animals. Although there is no information
from human studies, ganciclovir should be considered a potential carcinogen.
All HIV+ Patients: These patients may be receiving zidovudine (Retrovir?*). Patients should be counseled
that treatment with both ganciclovir and zidovudine simultaneously may not be tolerated by some patients
and may result in severe granulocytopenia (neutropenia). Patients with AIDS may be receiving didanosine
(Videx?#). Patients should be counseled that concomitant treatment with both ganciclovir and didanosine
can cause didanosine serum concentrations to be significantly increased.
HIV+ Patients With CMV Retinitis: Ganciclovir is not a cure for CMV retinitis, and immunocompromised
patients may continue to experience progression of retinitis during or following treatment. Patients
should be advised to have ophthalmologic follow-up examinations at a minimum of every 4 to 6 weeks while
being treated with CYTOVENE-IV or CYTOVENE. Some patients will require more frequent follow-up.
Transplant Recipients: Transplant recipients should be counseled regarding the high frequency of impaired
renal function in transplant recipients who received CYTOVENE-IV solution in controlled clinical trials,
particularly in patients receiving concomitant administration of nephrotoxic agents such as cyclosporine
and amphotericin B. Although the specific mechanism of this toxicity, which in most cases was reversible,
has not been determined, the higher rate of renal impairment in patients receiving CYTOVENE-IV solution
compared with those who received placebo in the same trials may indicate that CYTOVENE-IV played a
significant role.
(neutropenia), anemia and thrombocytopenia and that dose modifications may be required, including
discontinuation. The importance of close monitoring of blood counts while on therapy should be emphasized.
Patients should be informed that ganciclovir has been associated with elevations in serum creatinine.
Patients should be instructed to take CYTOVENE capsules with food to maximize bioavailability.
Patients should be advised that ganciclovir has caused decreased sperm production in animals and may cause
infertility in humans. Women of childbearing potential should be advised that ganciclovir causes birth
defects in animals and should not be used during pregnancy. Women of childbearing potential should be
advised to use effective contraception during treatment with CYTOVENE-IV or CYTOVENE. Similarly, men
should be advised to practice barrier contraception during and for at least 90 days following treatment
with CYTOVENE-IV or CYTOVENE.
Patients should be advised that ganciclovir causes tumors in animals. Although there is no information
from human studies, ganciclovir should be considered a potential carcinogen.
All HIV+ Patients: These patients may be receiving zidovudine (Retrovir?*). Patients should be counseled
that treatment with both ganciclovir and zidovudine simultaneously may not be tolerated by some patients
and may result in severe granulocytopenia (neutropenia). Patients with AIDS may be receiving didanosine
(Videx?#). Patients should be counseled that concomitant treatment with both ganciclovir and didanosine
can cause didanosine serum concentrations to be significantly increased.
HIV+ Patients With CMV Retinitis: Ganciclovir is not a cure for CMV retinitis, and immunocompromised
patients may continue to experience progression of retinitis during or following treatment. Patients
should be advised to have ophthalmologic follow-up examinations at a minimum of every 4 to 6 weeks while
being treated with CYTOVENE-IV or CYTOVENE. Some patients will require more frequent follow-up.
Transplant Recipients: Transplant recipients should be counseled regarding the high frequency of impaired
renal function in transplant recipients who received CYTOVENE-IV solution in controlled clinical trials,
particularly in patients receiving concomitant administration of nephrotoxic agents such as cyclosporine
and amphotericin B. Although the specific mechanism of this toxicity, which in most cases was reversible,
has not been determined, the higher rate of renal impairment in patients receiving CYTOVENE-IV solution
compared with those who received placebo in the same trials may indicate that CYTOVENE-IV played a
significant role.
GCV Atto interessato organismi
Herpes virus umano