Nizoral Cream




Categorie
Nizoral Cream Les marques, Nizoral Cream Analogs
Nizoral Cream Les marques melange
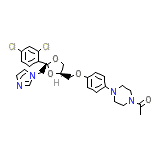
Nizoral Cream Formule chimique
C26H28Cl2N4O4
Nizoral Cream RX lien
http://www.rxlist.com/cgi/generic/ketocon.htm
Nizoral Cream FDA fiche
Nizoral Cream msds (fiche de securite des materiaux)
Nizoral Cream Synthese de reference
J. Heeres et al., US. 4,144,346 (1979)
Nizoral Cream Poids moleculaire
531.43 g/mol
Nizoral Cream Point de fusion
146 oC
Nizoral Cream H2O Solubilite
0,0866 mg / L
Nizoral Cream Etat
Solid
Nizoral Cream LogP
3.651
Nizoral Cream Formes pharmaceutiques
Comprimés; Shampooing; Crème; Suspension
Nizoral Cream Indication
Pour le traitement des éléments suivants infections fongiques systémiques: la candidose chronique candidose cutanéomuqueuse, muguet, candidurie, blastomycose, coccidioïdomycose, histoplasmose, chromomycose et paracoccidioidomycosis.
Nizoral Cream Pharmacologie
Le kétoconazole, comme le clotrimazole, le fluconazole, itraconazole, le miconazole et le, est un agent antifongique imidazole.
Nizoral Cream Absorption
Modérée
Nizoral Cream Toxicite
Hépatotoxicité, DL50 = 86 mg / kg (par voie orale chez le rat)
Nizoral Cream Information pour les patients
Shampoo: May be irritating to mucous membranes of the eyes and contact with this area should be avoided.
There have been reports that use of the shampoo resulted in removal of the curl from permanently waved hair.
Tablet: Patients should be instructed to report any signs and symptoms which may suggest liver dysfunction so that appropriate biochemical testing can be done. Such signs and symptoms may include unusual fatigue, anorexia, nausea and/or vomiting, jaundice, dark urine or pale stools
Nizoral Cream Organismes affectes
Champignons