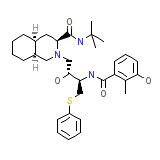
Mesylate




Categoria
Mesylate Nombres de marca, Mesylate Analogos
Mesylate Marca los nombres de mezcla
Mesylate Formula quimica
C32H45N3O4S
Mesylate RX enlace
http://www.rxlist.com/cgi/generic2/nelfin.htm
Mesylate FDA hoja
Mesylate MSDS (hoja de seguridad de materiales)
Mesylate Sintesis de referencia
No hay información disponible
Mesylate Peso molecular
567.784 g/mol
Mesylate Punto de fusion
349.84 oC
Mesylate H2O Solubilidad
Ligeramente soluble
Mesylate Estado
Solid
Mesylate LogP
5.247
Mesylate Formas de dosificacion
Tablet (oral - 250 mg, 625 mg) en polvo (oral)
Mesylate Indicacion
Mesylate Farmacologia
Nelfinavir es un inhibidor de la proteasa con actividad contra el virus de inmunodeficiencia humana tipo 1 (VIH-1). Inhibidores de la proteasa bloquean la parte del VIH llamada proteasa. VIH-1 proteasa es una enzima necesaria para la enzima. Esta inhibición impide la división de las poliproteínas viral que resulta en la formación de inmaduros no partículas virales infecciosas. Inhibidores de la proteasa se utilizan casi siempre en combinación
Mesylate Absorcion
Se absorbe bien tras la administración oral.
Mesylate Toxicidad
DL50 oral es más 5g/kg en ratas. Los efectos secundarios incluyen la sed y el hambre, la pérdida de peso inexplicable, aumento de la orina, fatiga, piel seca, picazón.
Mesylate Informacion de Pacientes
PATIENT INFORMATION
Patients should be informed that VIRACEPT is not a cure for HIV infection and that they may
continue to acquire illnesses associated with advanced HIV infection, including opportunistic
infections.
Patients should be told that the long-term effects of VIRACEPT are unknown at this time. They
should be told that there is currently no data demonstrating that VIRACEPT therapy can reduce
the risk of transmitting HIV to others through sexual contact or blood contamination.
Patients should be advised to take VIRACEPT every day as prescribed. Patients should not alter
the dose or discontinue therapy without consulting with their doctor. If a dose is missed, patients
should take the dose as soon as possible and then return to their normal schedule. However, if a dose
is skipped, the patient should not double the next dose.
The most frequent adverse event associated with VIRACEPT is diarrhea, which can usually be controlled
with non-prescription drugs, such as loperamide, which slow gastrointestinal motility.
VIRACEPT may interact with some drugs, therefore, patients should be advised to report to their doctor
the use of any other prescription or non-prescription medication.
Patients receiving oral contraceptives should be instructed that alternate or additional contraceptive
measures should be used during therapy with VIRACEPT.
Patients should be informed that VIRACEPT is not a cure for HIV infection and that they may
continue to acquire illnesses associated with advanced HIV infection, including opportunistic
infections.
Patients should be told that the long-term effects of VIRACEPT are unknown at this time. They
should be told that there is currently no data demonstrating that VIRACEPT therapy can reduce
the risk of transmitting HIV to others through sexual contact or blood contamination.
Patients should be advised to take VIRACEPT every day as prescribed. Patients should not alter
the dose or discontinue therapy without consulting with their doctor. If a dose is missed, patients
should take the dose as soon as possible and then return to their normal schedule. However, if a dose
is skipped, the patient should not double the next dose.
The most frequent adverse event associated with VIRACEPT is diarrhea, which can usually be controlled
with non-prescription drugs, such as loperamide, which slow gastrointestinal motility.
VIRACEPT may interact with some drugs, therefore, patients should be advised to report to their doctor
the use of any other prescription or non-prescription medication.
Patients receiving oral contraceptives should be instructed that alternate or additional contraceptive
measures should be used during therapy with VIRACEPT.
Mesylate Organismos afectados
Virus de inmunodeficiencia humana