Deflam




Categoria
Deflam Nombres de marca, Deflam Analogos
Deflam Marca los nombres de mezcla
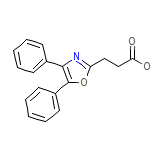
Deflam Formula quimica
Deflam RX enlace
Deflam FDA hoja
Deflam MSDS (hoja de seguridad de materiales)
Deflam Sintesis de referencia
Deflam Peso molecular
Deflam Punto de fusion
Deflam H2O Solubilidad
Deflam Estado
Deflam LogP
Deflam Formas de dosificacion
Deflam Indicacion
Deflam Farmacologia
Deflam Absorcion
Deflam Toxicidad
Deflam Informacion de Pacientes
Daypro, like other drugs of its class, can cause discomfort and, rarely, more serious side effects, such as gastrointestinal bleeding, which may result in hospitalization and even fatal outcomes. Although serious gastrointestinal tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms. Patients should be apprised of the importance of this follow-up.
Patients should report to their physicians the signs or symptoms of gastrointestinal ulceration or bleeding, skin rash, weight gain, or edema.
Patients should be informed of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, and “flu-like” symptoms). If these occur, patients should be instructed to stop therapy and seek immediate medical therapy.
Patients should also be instructed to seek immediate emergency help in the case of an anaphylactoid reaction.
In late pregnancy, as with other NSAIDs, Daypro should be avoided because it will cause premature closure of the ductus arteriosus.