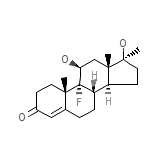
Ultandren




Ultandren Brand names, Ultandren Analogs
- Anadroid-F
- Androfluorene
- Androfluorone
- Android-f
- Androsterolo
- FXM
- Fluossimesterone [DCIT]
- Fluosterone
- Fluotestin
- Fluoximesterona [INN-Spanish]
- Fluoximesterone
- Fluoximesteronum
- Fluoxymesterone [BAN:INN:JAN]
- Fluoxymesteronum [INN-Latin]
- Fluoxymestrone
- Flusteron
- Flutestos
- Halodrin
- Halotestin
- Neo-Ormonal
- Ora Testryl
- Ora-testryl
- Oralsterone
- Oratestin
- Testoral
- Ultandren
- Ultandrene
Ultandren Brand Names Mixture
- Aldactazide 25 (Hydrochlorothiazide + Spironolactone)
- Aldactazide 50 (Hydrochlorothiazide + Spironolactone)
- Apo-Spirozide Tab (Hydrochlorothiazide + Spironolactone)
- Novo-Spirozine Tab 25mg (Hydrochlorothiazide + Spironolactone)
- Novo-Spirozine-50 Tab (Hydrochlorothiazide + Spironolactone)
Ultandren Chemical_Formula
C20H29FO3
Ultandren RX_link
http://www.rxlist.com/cgi/generic2/fluoxy.htm
Ultandren fda sheet
Ultandren msds (material safety sheet)
Ultandren Synthesis Reference
No information avaliable
Ultandren Molecular Weight
336.441 g/mol
Ultandren Melting Point
270 oC
Ultandren H2O Solubility
67.5 mg/L
Ultandren State
Solid
Ultandren LogP
2.537
Ultandren Dosage Forms
Tablet
Ultandren Indication
In males, used as replacement therapy in conditions associated with symptoms of deficiency or absence of endogenous testosterone. In females, for palliation of androgenresponsive recurrent mammary cancer in women who are more than one year but less than five years postmenopausal.
Ultandren Pharmacology
Fluoxymesterone is a synthetic androgen, or male hormone, similar to testosterone. Fluoxymesterone works by attaching itself to androgen receptors; this causes it to interact with the parts of the cell involved in the making of proteins. It may cause an increase in the synthesis of some proteins or a decrease in the synthesis of others. These proteins have a variety of effects, including blocking the growth of some types of breast cancer cells, stimulating cells that cause male sexual characteristics, and stimulating the production of red blood cells.
Ultandren Absorption
Oral absorption is less than 44%.
Ultandren side effects and Toxicity
Side effects include virilization (masculine traits in women), acne, fluid retention, and hypercalcemia.
Ultandren Patient Information
Ultandren Organisms Affected
Humans and other mammals