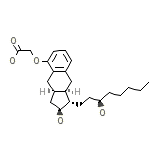
Treprostinil




Treprostinil Brand names, Treprostinil Analogs
Treprostinil Brand Names Mixture
- No information avaliable
Treprostinil Chemical_Formula
C23H34O5
Treprostinil RX_link
http://www.rxlist.com/cgi/generic/remodulin.htm
Treprostinil fda sheet
Treprostinil msds (material safety sheet)
Treprostinil Synthesis Reference
No information avaliable
Treprostinil Molecular Weight
390.513 g/mol
Treprostinil Melting Point
No information avaliable
Treprostinil H2O Solubility
Insoluble at 25°C
Treprostinil State
Solid
Treprostinil LogP
4.08
Treprostinil Dosage Forms
Solution for injection (supplied in 20 mL multi-use vials in four strengths, containing 1 mg/mL, 2.5 mg/mL, 5 mg/mL or 10 mg/mL of treprostinil)
Treprostinil Indication
For use as a continuous subcutaneous infusion or intravenous infusion (for those not able to tolerate a subcutaneous infusion) for the treatment of pulmonary arterial hypertension in patients with NYHA Class II-IV symptoms to diminish symptoms associated with exercise.
Treprostinil Pharmacology
Pulmonary arterial hypertension (PAH) is a disease in which blood pressure is abnormally high in the arteries between the heart and lungs. PAH is characterized by symptoms of shortness of breath during physical exertion. The condition can ultimately lead to heart failure. Treprostinil is a potent oral antiplatelet agent. The major pharmacologic actions of treprostinil are direct vasodilation of pulmonary and systemic arterial vascular beds and inhibition of platelet aggregation. In animals, the vasodilatory effects reduce right and left ventricular afterload and increase cardiac output and stroke volume. Other studies have shown that treprostinil causes a dose-related negative inotropic and lusitropic effect. No major effects on cardiac conduction have been observed.
Treprostinil Absorption
Relatively rapid and complete after subcutaneous infusion, with an absolute bioavailability approximately 100%. In patients with mild (n=4) or moderate (n=5) hepatic insufficiency and portopulmonary hypertension following a subcutaneous dose of 10 ng per kg of body weight per min for 150 mins the AUC 0-∞ was increased 3-fold and 5-fold respectively.
Treprostinil side effects and Toxicity
Symptoms of overdose are extensions of its dose-limiting pharmacologic effects and include flushing, headache, hypotension, nausea, vomiting, and diarrhea. Most events were self-limiting and resolved with reduction or withholding of treprostinil.
Treprostinil Patient Information
Treprostinil Organisms Affected
Humans and other mammals