Sevelamer




Sevelamer Brand names, Sevelamer Analogs
Sevelamer Brand Names Mixture
- No information avaliable
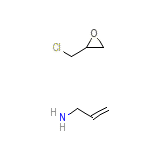
Sevelamer Chemical_Formula
C6H12ClNO
Sevelamer RX_link
http://www.rxlist.com/cgi/generic2/sevel.htm
Sevelamer fda sheet
Sevelamer msds (material safety sheet)
Sevelamer Synthesis Reference
No information avaliable
Sevelamer Molecular Weight
149.618 g/mol
Sevelamer Melting Point
No information avaliable
Sevelamer H2O Solubility
Insoluble
Sevelamer State
Solid
Sevelamer LogP
0.559
Sevelamer Dosage Forms
Capsules and film-coated tablets (800 mg or 400 mg)
Sevelamer Indication
For the control of serum phosphorus in patients with Chronic Kidney Disease (CKD) on hemodialysis.
Sevelamer Pharmacology
Patients with end-stage renal disease (ESRD) retain phosphorus and can develop hyperphosphatemia. High serum phosphorus can precipitate serum calcium resulting in ectopic calcification. When the product of serum calcium and phosphorus concentrations (Ca x P) exceeds 55 mg2/dL2, there is an increased risk that ectopic calcification will occur. Hyperphosphatemia plays a role in the development of secondary hyperparathyroidism in renal insufficiency. An increase in parathyroid hormone (PTH) levels is characteristic of patients with chronic renal failure. Increased levels of PTH can lead to osteitis fibrosa, a bone disease. A decrease in serum phosphorus may decrease serum PTH levels. Treatment of hyperphosphatemia includes reduction in dietary intake of phosphate, inhibition of intestinal phosphate absorption with phosphate binders, and removal of phosphate with dialysis. Sevelamer taken with meals has been shown to decrease serum phosphorus concentrations in patients with ESRD who are on hemodialysis. In vitro studies have shown that the capsule and tablet formulations bind phosphate to a similar extent. Sevelamer treatment also results in a lowering of low-density lipoprotein (LDL) and total serum cholesterol levels.
Sevelamer Absorption
Not absorbed following oral administration, however no absorption studies have been performed in patients with renal disease.
Sevelamer side effects and Toxicity
Sevelamer has been given to normal healthy volunteers in doses of up to 14 grams per day for eight days with no adverse effects. Sevelamer has been given in average doses up to 13 grams per day to hemodialysis patients. There are no reported overdosages of sevelamer in patients. Since sevelamer is not absorbed, the risk of systemic toxicity is low.
Sevelamer Patient Information
No information avaliable
Sevelamer Organisms Affected
Humans and other mammals