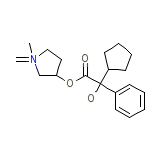
Robanul




Robanul Brand names, Robanul Analogs
Robanul Brand Names Mixture
- No information avaliable
Robanul Chemical_Formula
C19H28NO3+
Robanul RX_link
http://www.rxlist.com/cgi/generic3/glycopyrrolate.htm
Robanul fda sheet
Robanul msds (material safety sheet)
Robanul Synthesis Reference
No information avaliable
Robanul Molecular Weight
318.431 g/mol
Robanul Melting Point
192.5 oC
Robanul H2O Solubility
No information avaliable
Robanul State
Solid
Robanul LogP
-0.99
Robanul Dosage Forms
Liquid; Tablet
Robanul Indication
For use as a preoperative antimuscarinic to reduce salivary, tracheobronchial, and pharyngeal secretions, to reduce the volume and free acidity of gastric secretions and to block cardiac vagal inhibitory reflexes during induction of anesthesia and intubation.
Robanul Pharmacology
Glycopyrrolate decreases acid secretion in the stomach. Hence it can be used for treating ulcers in the stomach and small intestine, in combination with other medications. In anesthesia, glycopyrrolate injection serves as a preoperative antimuscarinic operation that reduces salivary, tracheobronchial, and pharyngeal secretions, as well as decreases the acidity of gastric secretions blocks cardiac vagal inhibitory reflexes during intubation
Robanul Absorption
Rapidly absorbed (1-2 minutes) after intravenous injection
Robanul side effects and Toxicity
Side effects include dry mouth, difficult urinating, heachaches, diarrhea and constipation. The medication also induces drowsiness or blurred vision. LD50=709 mg/kg (rat, oral).
Robanul Patient Information
No information avaliable
Robanul Organisms Affected
Humans and other mammals