Novo-Timol




Novo-Timol Brand names, Novo-Timol Analogs
Novo-Timol Brand Names Mixture
- Combigan (Brimonidine Tartrate + Timolol Maleate)
- Cosopt -(2%/0.5% Oph Dps) (Dorzolamide Hydrochloride + Timolol Maleate)
- Timolide Tab (Hydrochlorothiazide + Timolol Maleate)
- Timpilo 2 (Pilocarpine Hydrochloride + Timolol Maleate)
- Timpilo 4 (Pilocarpine Hydrochloride + Timolol Maleate)
- Xalacom (Latanoprost + Timolol Maleate)
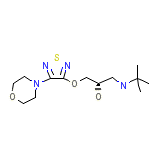
Novo-Timol Chemical_Formula
Novo-Timol RX_link
Novo-Timol fda sheet
Novo-Timol msds (material safety sheet)
Novo-Timol Synthesis Reference
Novo-Timol Molecular Weight
Novo-Timol Melting Point
Novo-Timol H2O Solubility
Novo-Timol State
Novo-Timol LogP
Novo-Timol Dosage Forms
Novo-Timol Indication
Novo-Timol Pharmacology
Novo-Timol Absorption
Novo-Timol side effects and Toxicity
Novo-Timol Patient Information
Patients should be instructed to avoid allowing the tip of the dispensing container to contact the eye or surrounding structures. Patients should also be instructed that ocular solutions, if handled improperly, could become contaminated by common bacteria known to cause ocular infections. Serious damage to the eye and subsequent loss of vision may result from using contaminated solutions.
Patients should also be advised that if they have ocular surgery or develop an intercurrent ocular condition (e.g.,trauma or infection), they should immediately seek their physicianís advice concerning the continued use of the present multidose container. Patients should be instructed to invert the closed container and shake once before each use. It is not necessary to shake the container more than once. Patients requiring concomitant topical ophthalmic medications should be instructed to administer these at least 10 minutes before instilling Timolol GFS. Patients with bronchial asthma, a history of bronchial asthma, severe chronic obstructive pulmonary disease, sinus bradycardia, second or third degree atrioventricular block, or cardiac failure should be advised not to take this product. Transient blurred vision or visual disturbance, generally lasting from 30 seconds to 5 minutes, following instillation may impair the ability to perform hazardous tasks such as operating machinery or driving a motor vehicle.