Merrem I.V.




Merrem I.V. Brand names, Merrem I.V. Analogs
Merrem I.V. Brand Names Mixture
- No information avaliable
Merrem I.V. Chemical_Formula
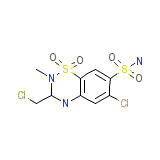
C9H11Cl2N3O4S2
Merrem I.V. RX_link
http://www.rxlist.com/cgi/generic3/methyclothiazide.htm
Merrem I.V. fda sheet
Merrem I.V. msds (material safety sheet)
Merrem I.V. Synthesis Reference
No information avaliable
Merrem I.V. Molecular Weight
360.239 g/mol
Merrem I.V. Melting Point
225 oC
Merrem I.V. H2O Solubility
11.2 mg/L
Merrem I.V. State
Solid
Merrem I.V. LogP
0.604
Merrem I.V. Dosage Forms
Tablet for oral administration (5 mg)
Merrem I.V. Indication
For use in the management of hypertension either as the sole therapeutic agent or to enhance the effect of other antihypertensive drugs in the more severe forms of hypertension. Also used as adjunctive therapy in edema associated with congestive heart failure, hepatic cirrhosis, and corticosteroid and estrogen therapy.
Merrem I.V. Pharmacology
Methyclothiazide, a diuretic-antihypertensive agent, is a member of the benzothiadiazine (thiazide) class of drugs. Methyclothiazide has a per mg natriuretic activity approximately 100 times that of the prototype thiazide, chlorothiazide. At maximal therapeutic dosages, all thiazides are approximately equal in their diuretic/natriuretic effects. Like other benzothiadiazines, methyclothiazide also has antihypertensive properties, and may be used for this purpose either alone or to enhance the antihypertensive action of other drugs.
Merrem I.V. Absorption
Rapidly absorbed following oral administration.
Merrem I.V. side effects and Toxicity
Acute oral toxicity (LD50): >4000 mg/kg [Rat]. Symptoms of overdosage include electrolyte imbalance and signs of potassium deficiency such as confusion, dizziness, muscular weakness, and gastrointestinal disturbances.
Merrem I.V. Patient Information
No information avaliable
Merrem I.V. Organisms Affected
Humans and other mammals