MK 996




MK 996 Brand names, MK 996 Analogs
MK 996 Brand Names Mixture
- No information avaliable
MK 996 Chemical_Formula
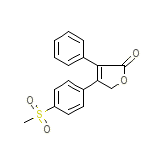
C17H14O4S
MK 996 RX_link
http://www.rxlist.com/cgi/generic/rofecox.htm
MK 996 fda sheet
MK 996 msds (material safety sheet)
MK 996 Synthesis Reference
No information avaliable
MK 996 Molecular Weight
314.357 g/mol
MK 996 Melting Point
No information avaliable
MK 996 H2O Solubility
Insoluble
MK 996 State
Solid
MK 996 LogP
3.019
MK 996 Dosage Forms
Tablet (12.5 mg, 25 mg, or 50 mg); Oral suspension (12.5 or 25 mg per 5 mL solution)
MK 996 Indication
For the treatment of osteoarthritis, acute pain in adults and menstrual pain.
MK 996 Pharmacology
Rofecoxib, a selective cyclooxygenase-2 (COX-2) inhibitor, is classified as a nonsteroidal anti-inflammatory drug (NSAID). Rofecoxib is used for its anti-inflammatory, analgesic, and antipyretic activities in the management of osteoarthritis (OA) and for the treatment of dysmenorrhea or acute pain. Unlike celecoxib, rofecoxib lacks a sulfonamide chain and does not require CYP450 enzymes for metabolism.
MK 996 Absorption
The mean oral bioavailability of rofecoxib at therapeutically recommended doses of 12.5, 25, and 50 mg is approximately 93%.
MK 996 side effects and Toxicity
No overdoses of rofecoxib were reported during clinical trials. Administration of single doses of rofecoxib 1000 mg to 6 healthy volunteers and multiple doses of 250 mg/day for 14 days to 75 healthy volunteers did not result in serious toxicity.
MK 996 Patient Information
PATIENT INFORMATION
Physicians should instruct their patients to read the patient package insert before starting therapy with VIOXX and to reread
it each time the prescription is renewed in case any information has changed.
VIOXX can cause discomfort and, rarely, more serious side effects, such as gastrointestinal bleeding, which may result in
hospitalization and even fatal outcomes. Although serious GI tract ulcerations and bleeding can occur without warning symptoms,
patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing
any indicative signs or symptoms. Patients should be apprised of the importance of this follow-up. Risk of GI Ulceration, Bleeding
and Perforation. Patients should be informed that VIOXX is not a substitute for aspirin for cardiovascular prophylaxis because of
its lack of effect on platelets. For additional cardiovascular safety information see CLINICAL STUDIES, Special Studies, VIGOR and
PRECAUTIONS, Cardiovascular Effects.
Patients should promptly report signs or symptoms of gastrointestinal ulceration or bleeding, skin rash, unexplained weight gain,
edema or chest pain to their physicians.
Physicians should instruct their patients to read the patient package insert before starting therapy with VIOXX and to reread
it each time the prescription is renewed in case any information has changed.
VIOXX can cause discomfort and, rarely, more serious side effects, such as gastrointestinal bleeding, which may result in
hospitalization and even fatal outcomes. Although serious GI tract ulcerations and bleeding can occur without warning symptoms,
patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing
any indicative signs or symptoms. Patients should be apprised of the importance of this follow-up. Risk of GI Ulceration, Bleeding
and Perforation. Patients should be informed that VIOXX is not a substitute for aspirin for cardiovascular prophylaxis because of
its lack of effect on platelets. For additional cardiovascular safety information see CLINICAL STUDIES, Special Studies, VIGOR and
PRECAUTIONS, Cardiovascular Effects.
Patients should promptly report signs or symptoms of gastrointestinal ulceration or bleeding, skin rash, unexplained weight gain,
edema or chest pain to their physicians.
MK 996 Organisms Affected
Humans and other mammals