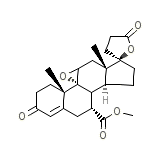
INSPRA




INSPRA Brand names, INSPRA Analogs
INSPRA Brand Names Mixture
- No information avaliable
INSPRA Chemical_Formula
C24H30O6
INSPRA RX_link
http://www.rxlist.com/cgi/generic/inspra.htm
INSPRA fda sheet
INSPRA msds (material safety sheet)
INSPRA Synthesis Reference
No information avaliable
INSPRA Molecular Weight
414.491 g/mol
INSPRA Melting Point
No information avaliable
INSPRA H2O Solubility
Slightly soluble
INSPRA State
Solid
INSPRA LogP
2.445
INSPRA Dosage Forms
Tablet (film-coated - 25, 50 mg)
INSPRA Indication
For improvement of survival of stable patients with left ventricular systolic dysfunction (ejection fraction <40%) and clinical evidence of congestive heart failure after an acute myocardial infarction.
INSPRA Pharmacology
Eplerenone, an aldosterone receptor antagonist similar to spironolactone, has been shown to produce sustained increases in plasma renin and serum aldosterone, consistent with inhibition of the negative regulatory feedback of aldosterone on renin secretion. The resulting increased plasma renin activity and aldosterone circulating levels do not overcome the effects of eplerenone. Eplerenone selectively binds to recombinant human mineralocorticoid receptors relative to its binding to recombinant human glucocorticoid, progesterone and androgen receptors.
INSPRA Absorption
The absolute bioavailability of eplerenone is unknown.
INSPRA side effects and Toxicity
The most likely symptoms of human overdosage would be anticipated to be hypotension or hyperkalemia. However, no cases of human overdosage with eplerenone have been reported.
INSPRA Patient Information
INSPRA Organisms Affected
Humans and other mammals