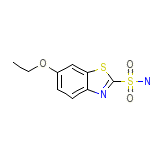
Glaucotensil




Glaucotensil Brand names, Glaucotensil Analogs
Glaucotensil Brand Names Mixture
- No information avaliable
Glaucotensil Chemical_Formula
C9H10N2O3S2
Glaucotensil RX_link
No information avaliable
Glaucotensil fda sheet
Glaucotensil msds (material safety sheet)
Glaucotensil Synthesis Reference
No information avaliable
Glaucotensil Molecular Weight
258.319 g/mol
Glaucotensil Melting Point
189 oC
Glaucotensil H2O Solubility
40 mg/L
Glaucotensil State
Solid
Glaucotensil LogP
1.218
Glaucotensil Dosage Forms
Tablet (125, 250 mg); Capsule (500mg); Injection (500mg)
Glaucotensil Indication
For use in the treatment of duodenal ulcers, as a diuretic, and in the treatment of glaucoma, and may also be useful in the treatment of seizures associated with epilepsy.
Glaucotensil Pharmacology
Ethoxzolamide, a sulfonamide, inhibits carbonic anhydrase activity in proximal renal tubules to decrease reabsorption of water, sodium, potassium, bicarbonate. It also decreases carbonic anhydrase in the CNS, increasing the seizure threshold. This reduction in carbonic anhydrase also reduces the intraocular pressure in the eye by decreasing aqueous humor.
Glaucotensil Absorption
Rapidly absorbed with 65% bioavailability
Glaucotensil side effects and Toxicity
No information avaliable
Glaucotensil Patient Information
Glaucotensil Organisms Affected
Humans and other mammals