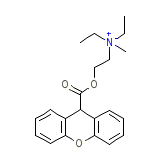
Gastrin I




Gastrin I Brand names, Gastrin I Analogs
- Asabaine
- Avagal
- Banthin
- Banthine
- Banthine Bromide
- Dixamone Bromide
- Doladene
- Frenogastrico
- Gastrin I
- Gastrin-1 Human
- Gastron
- Gastrosedan
- MTB 51
- Mantheline
- Metantyl
- Metaxan
- Methanide
- Methantheline Bromide
- Methanthelinium
- Methanthelinium Bromide
- Methanthelinum
- Methanthine Bromide
- Methelina
- Resobantin
- Ulcine
- Ulcudexter
- Vagamin
- Vagantin
- Xanteline
Gastrin I Brand Names Mixture
- No information avaliable
Gastrin I Chemical_Formula
C21H26NO3
Gastrin I RX_link
No information avaliable
Gastrin I fda sheet
Gastrin I msds (material safety sheet)
Gastrin I Synthesis Reference
No information avaliable
Gastrin I Molecular Weight
340.436 g/mol
Gastrin I Melting Point
No information avaliable
Gastrin I H2O Solubility
No information avaliable
Gastrin I State
Solid
Gastrin I LogP
No information avaliable
Gastrin I Dosage Forms
Tablet (50 mg)
Gastrin I Indication
For the treatment of peptic ulcer disease, irritable bowel syndrome, pancreatitis, gastritis, biliary dyskinesia, pylorosplasm, and reflex neurogenic bladder in children.
Gastrin I Pharmacology
Methantheline is a synthetic quarternary ammonium antimuscarinic used to relieve cramps or spasms of the stomach, intestines, and bladder. It can be used together with antacids or other medicines, such as H2-receptor antagonists, in the treatment of peptic ulcer. Methantheline inhibits muscarinic actions at postganglionic parasympathetic neuroeffector sites.
Gastrin I Absorption
Rapidly absorbed.
Gastrin I side effects and Toxicity
Symptoms of overdose: blurred vision (continuing) or changes in near vision, clumsiness or unsteadiness, confusion, convulsions, difficulty in breathing, muscle weakness (severe), or tiredness (severe), dizziness, drowsiness (severe), dryness of mouth, nose, or throat (severe), fast heartbeat, fever, hallucinations, slurred speech, unusual excitement, nervousness, restlessness, or irritability, unusual warmth, dryness, and flushing of skin.
Gastrin I Patient Information
No information avaliable
Gastrin I Organisms Affected
Humans and other mammals