FK5




FK5 Brand names, FK5 Analogs
FK5 Brand Names Mixture
- No information avaliable
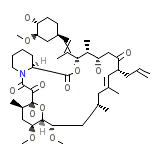
FK5 Chemical_Formula
FK5 RX_link
FK5 fda sheet
FK5 msds (material safety sheet)
FK5 Synthesis Reference
FK5 Molecular Weight
FK5 Melting Point
FK5 H2O Solubility
FK5 State
FK5 LogP
FK5 Dosage Forms
FK5 Indication
FK5 Pharmacology
FK5 Absorption
FK5 side effects and Toxicity
FK5 Patient Information
Read this important information before you start using PROTOPIC [pro-TOP-ik] Ointment and each time you refill your prescription. There may be new information. This summary is not meant to take the place of your doctorís advice.
What Is PROTOPIC?
PROTOPIC Ointment is a prescription medicine that is used to treat eczema (atopic dermatitis). It is for adults and
children age 2 years and older. You can use PROTOPIC for short or intermittent long periods of treatment.
Intermittent means starting and stopping repeatedly, as directed by your doctor. You can use it on all affected areas
of your skin, including your face and neck.
Who should not use PROTOPIC? Do not use PROTOPIC if you are
ï breastfeeding
ï allergic to PROTOPIC Ointment or any of its ingredients. The active ingredient is tacrolimus. Ask your doctor or pharmacist about the inactive ingredients.
Before you start using PROTOPIC, tell your doctor if you are:
ï using any other prescription medicines, non-prescription (over-the-counter) medicines, or supplements
ï receiving any form of light therapy (phototherapy, UVA or UVB) on your skin
ï using any other type of skin product ï pregnant or planning to become pregnant
How Do I Use PROTOPIC?
Use PROTOPIC only to treat eczema that has been diagnosed by a doctor.
ï Wash your hands before using PROTOPIC.
ï Apply a thin layer of PROTOPIC to all skin areas that your doctor has diagnosed as eczema. Try to cover the affected areas completely. Most people find that a pea-sized amount squeezed from the tube covers an area about the size of a two-inch circle (approximately the size of a silver dollar).
ï Apply the ointment twice a day, about 12 hours apart.
ï Before applying PROTOPIC Ointment after a bath or shower, be sure your skin is completely dry.
ï Do not cover the skin being treated with bandages, dressings or wraps. Unless otherwise instructed by your doctor, do not apply another type of skin product on top of PROTOPIC Ointment. However, you can wear normal clothing.
ï Do not bathe, shower or swim right after applying PROTOPIC. This could wash off the ointment.
ï If you are a caregiver applying PROTOPIC Ointment to a patient, or if you are a patient who is not treating your hands, wash your hands with soap and water after applying PROTOPIC. This should remove any ointment left on the hands.
ï Use PROTOPIC only on your skin. Do not swallow PROTOPIC.
Because 2 strengths of PROTOPIC are available for adult patients, your doctor will decide what strength of PROTOPIC Ointment is best for you.
Many people notice that their skin starts to improve after the first few weeks of treatment. Even though your skin looks and feels better, it is important to keep using PROTOPIC as instructed by your doctor.
If you do not notice an improvement in your eczema or if your eczema gets worse within the first few weeks of treatment, tell your doctor.
What Should I Avoid While Using PROTOPIC?
ï Avoid sunlight and sun lamps, tanning beds, and treatment with UVA or UVB light. If you need to be outdoors after applying PROTOPIC, wear loose fitting clothing that protects the treated area from the sun. In addition, ask your doctor what other type of protection from the sun you should use.
ï Check with your doctor or pharmacist before you
ï start taking any new medicines while using PROTOPIC.
ï Start using any other ointment, lotions, or creams on your skin.
What Are The Possible Side Effects of PROTOPIC?
The most common side effects of PROTOPIC are stinging, soreness, a burning feeling, or itching of the skin treated with PROTOPIC. These side effects are usually mild to moderate, are most common during the first few days of treatment, and typically lessen if your skin heals.
Less common side effects include acne, swollen or infected hair follicles, headache, increased sensitivity of the skin to hot or cold temperatures, or flu-like symptoms [common cold and congestion (stuffy nose)]. Some people may get skin tingling, upset stomach, herpes zoster (chicken pox or shingles), or muscle pain. While you are using PROTOPIC, drinking alcohol may cause the skin or face to become flushed or red and feel hot. Call your doctor if side effects continue or become a problem.
How Should I Store PROTOPIC?
Store PROTOPIC at room temperature (59° to 86°F). For instance, never leave PROTOPIC in your car in cold or hot weather. Make sure the cap on the tube is tightly closed. Keep PROTOPIC out of the reach of children.
General Advice about Prescription Medicines
Do not use PROTOPIC for a condition for which it was not prescribed. If you have any concerns about PROTOPIC, ask your doctor. Your doctor or pharmacist can give you information about PROTOPIC that was written for health care professionals. For more information, you can also visit the Fujisawa Internet site at www.fujisawa.com or call the PROTOPIC Help Line at 1-800-727-7003.