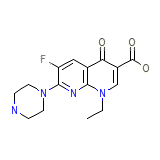
Echodide




Echodide Brand names, Echodide Analogs
Echodide Brand Names Mixture
- No information avaliable
Echodide Chemical_Formula
C15H17FN4O3
Echodide RX_link
http://www.rxlist.com/cgi/generic2/enoxacin.htm
Echodide fda sheet
Echodide msds (material safety sheet)
Echodide Synthesis Reference
No information avaliable
Echodide Molecular Weight
320.319 g/mol
Echodide Melting Point
220-224 oC
Echodide H2O Solubility
3.43 g/L
Echodide State
Solid
Echodide LogP
0.387
Echodide Dosage Forms
200 mg and 400 mg film-coated tablets
Echodide Indication
For the treatment of adults (≥18 years of age) with the following infections caused by susceptible strains of the designated microorganisms: (1) uncomplicated urethral or cervical gonorrhea due to Neisseria gonorrhoeae, (2) uncomplicated urinary tract infections (cystitis) due to Escherichia coli, Staphylococcus epidermidis, or Staphylococcus saprophyticus, and (3) complicated urinary tract infections due to Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis, Pseudomonas aeruginosa, Staphylococcus epidermidis, or Enterobacter cloacae.
Echodide Pharmacology
Enoxacin is a quinolone/fluoroquinolone antibiotic. Enoxacin is bactericidal and its mode of action depends on blocking of bacterial DNA replication by binding itself to an enzyme called DNA gyrase, which allows the untwisting required to replicate one DNA double helix into two. Enoxacin is a broad-spectrum antibiotic that is active against both Gram-positive and Gram-negative bacteria. Enoxacin may be active against pathogens resistant to drugs that act by different mechanisms.
Echodide Absorption
Rapidly absorbed following oral administration, with an absolute oral bioavailability of approximately 90%.
Echodide side effects and Toxicity
No information avaliable
Echodide Patient Information
Echodide Organisms Affected
Enteric bacteria and other eubacteria