DUP 89




DUP 89 Brand names, DUP 89 Analogs
DUP 89 Brand Names Mixture
- Hyzaar
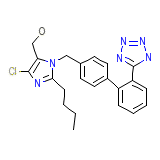
DUP 89 Chemical_Formula
C22H23ClN6O
DUP 89 RX_link
http://www.rxlist.com/cgi/generic/losar.htm
DUP 89 fda sheet
DUP 89 msds (material safety sheet)
DUP 89 Synthesis Reference
D. J. Carini et al., U.S. Pat. 5,138,69 (1992)
DUP 89 Molecular Weight
422.911 g/mol
DUP 89 Melting Point
183.5-184.5 oC
DUP 89 H2O Solubility
0.82 mg/L
DUP 89 State
Solid
DUP 89 LogP
5.438
DUP 89 Dosage Forms
Tablet
DUP 89 Indication
For the treatment of hypertension.
DUP 89 Pharmacology
Losartan is the first of a class of antihypertensive agents called angiotensin II (AG II) receptor antagonists. It is, along with its longer acting active metabolite (E-3174), a specific and selective AT1 receptor antagonist. Losartan blocks the vasoconstrictor and aldosterone-secreting effects of angiotensin II by selectively blocking the binding of angiotensin II to the AT1 receptor found in many tissues, (e.g., vascular smooth muscle, adrenal gland).
DUP 89 Absorption
Well absorbed, the systemic bioavailability of losartan is approximately 33%
DUP 89 side effects and Toxicity
Hypotension and tachycardia; Bradycardia could occur from parasympathetic (vagal) stimulation, LD50= 1000 mg/kg (orally in rat)
DUP 89 Patient Information
PATIENT INFORMATION
Losartan potassium is used to treat high blood pressure. It may be used alone
or with other medications for high blood pressure. This medication should,
generally, not be used during pregnancy. If you are pregnant or considering
becoming pregnant, inform your physician immediately to discuss the risks
versus benefits. There are few side effects associated with losartan potassium.
Those most frequently reported, cough, upper respiratory infection and
dizziness, occurred no more frequently than in patients who did not take this
medication. You may take this medication with or without food. You should have
your blood pressure checked regularly
Losartan potassium is used to treat high blood pressure. It may be used alone
or with other medications for high blood pressure. This medication should,
generally, not be used during pregnancy. If you are pregnant or considering
becoming pregnant, inform your physician immediately to discuss the risks
versus benefits. There are few side effects associated with losartan potassium.
Those most frequently reported, cough, upper respiratory infection and
dizziness, occurred no more frequently than in patients who did not take this
medication. You may take this medication with or without food. You should have
your blood pressure checked regularly
DUP 89 Organisms Affected
Humans and other mammals